2.1: Chemistry Tutorial
- Page ID
- 48546
2.1 Chemistry Tutorial
The chemical compounds that are important for understanding most of the chemistry in this course are organic - that means that the compounds primarily contain carbon, hydrogen, and oxygen atoms (also sulfur and nitrogen). They can also be called hydrocarbons. The basic structures that we will be discussing in this course are called: 1) alkane (aka aliphatic), 2) branched alkane, 3) cycloalkane, 4) alkenes (double-bonds), 5) aromatic, 6) hydroaromatic, and 7) alcohols. First, I will show the atoms and how they are connected using the element abbreviation and lines as bonds, and then I will show abbreviated structural representations.
1. Alkane - atoms are lined up. For stick representation, each corner represents a CH2 group, and each end represents a CH3 group.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| Heptane (7 C atoms) | 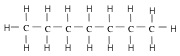 |
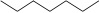 |
2. Branched Alkane - still an alkane, but instead of a straight line, the carbons are branched off of each other.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| Isobutane (4 C atoms) | 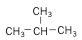 |
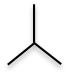 |
| Isopentane (5 C atoms) | 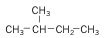 |
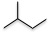 |
3. Cycloalkanes - again, still an alkane, but forms a ring compound.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| Cyclohexane (6 C atoms) | 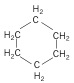 |
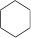 |
4. Alkenes - alkanes that contain a double bond.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| Pentene (5 C atoms) | 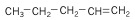 |
 |
5. Aromatic - hydrocarbon ring compound with single and double bonds, significant differences in properties.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| Benzene (6 C atoms) | 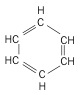 |
 , ,  |
6. Hydroaromatics - hydrocarbon ring compound with an aromatic and an alkane in one molecule.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| 1,2,3,4-tetrahydronaphthalene, aka tetralin (10 C atoms) | 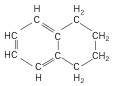 |
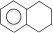 |
7. Alcohols - hydrocarbon with -OH functional group.
| Name | Atoms and Bonds | Stick Representation |
|---|---|---|
| Butanol (4 C atoms) | 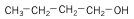 |
 |
| Ethanol (2 C atoms) | 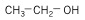 |
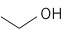 |
The following table shows common hydrocarbons and their properties. It is important to know the properties of various hydrocarbons so that we can separate them and make chemical changes to them. This is a very brief overview - we will not yet be going into significant depth as to why the differences in chemicals affect the properties.
Table 2.1: List of Common Hydrocarbons and Properties
| Name | Number of C Atoms | Molecular Formula |
bp (°C), 1 atm |
mp (°C) |
Density (g/mL) (@20°C) |
|---|---|---|---|---|---|
| Methane | 1 | CH4 | -161.5 | -182 | -- |
| Ethane | 2 | C2H6 | -88.6 | -183 | -- |
| Propane | 3 | C3H8 | -42.1 | -188 | -- |
| Butane | 4 | C4H10 | -0.5 | -138 | -- |
| Pentane | 5 | C5H12 | 36.1 | -130 | 0.626 |
| Hexane | 6 | C6H14 | 68.7 | -95 | 0.659 |
| Heptane | 7 | C7H16 | 98.4 | -91 | 0.684 |
| Octane | 8 | C8H18 | 125.7 | -57 | 0.703 |
| Nonane | 9 | C9H20 | 150.8 | -54 | 0.718 |
| Decane | 10 | C10H22 | 174.1 | -30 | 0.730 |
| Tetradecane | 14 | C14H30 | 253.5 | 6 | 0.763 |
| Hexadecane | 16 | C16H34 | 287 | 18 | 0.770 |
| Heptadecane | 17 | C17H36 | 303 | 22 | 0.778 |
| Eicosane | 20 | C20H42 | 343 | 36.8 | 0.789 |
| Cyclohexane | 6 | C6H12 | 81 | 6.5 | 0.779 |
| Cyclopentane | 5 | C5H10 | 49 | -94 | 0.751 |
| Ethanol | 2 | C2H6O | 78 | -114 | 0.789 |
| Butanol | 4 | C4H10O | 118 | -90 | 0.810 |
| Pentene | 5 | C5H10 | 30 | -165 | 0.640 |
| Hexene | 6 | C6H12 | 63 | -140 | 0.673 |
| Benzene | 6 | C6H6 | 80.1 | 5.5 | 0.877 |
| Naphthalene | 10 | C10H8 | 218 | 80 | 1.140 |
| 1,2,3,4-Tetrahydronaphthalene | 10 | C10H12 | 207 | -35.8 | 0.970 |


