4.3.2.2: Pressure Measurement
- Page ID
- 664
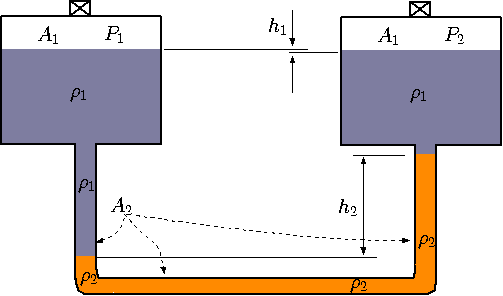
Fig. 4.7 Schematic of sensitive measurement device.
The idea describes the atmospheric measurement that can be extended to measure the pressure of the gas chambers. Consider a chamber filled with gas needed to be measured (see Figure 4.6). One technique is to attached ``U'' tube to the chamber and measure the pressure. This way, the gas is prevented from escaping and its pressure can be measured with a minimal interference to the gas (some gas enters to the tube). The gas density is significantly lower than the liquid density and therefore can be neglected. The pressure at point "1'' is
\[
P_1 = P_{atmos} + \rho\, g \, h
\label{static:eq:gageP}
\]
Contributors and Attributions
Dr. Genick Bar-Meir. Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or later or Potto license.


