4.3.4.2 The Stability Analysis
- Page ID
- 671
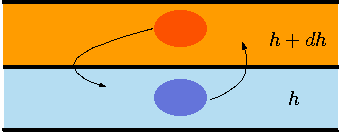
Fig. 4.11 Two adjoin layers for stability analysis.
It is interesting to study whether this solution (98) is stable and if so under what conditions. Suppose that for some reason, a small slab of material moves from a layer at height, \(h\), to layer at height \(h+dh\) (see Figure 4.11) What could happen? There are two main possibilities one: the slab could return to the original layer or two: stay at the new layer (or even move further, higher heights). The first case is referred to as the stable condition and the second case referred to as the unstable condition. The whole system falls apart and does not stay if the analysis predicts unstable conditions. A weak wind or other disturbances can make the unstable system to move to a new condition. This question is determined by the net forces acting on the slab. Whether these forces are toward the original layer or not. The two forces that act on the slab are the gravity force and the surroundings pressure (buoyant forces). Clearly, the slab is in equilibrium with its surroundings before the movement (not necessarily stable). Under equilibrium, the body forces that acting on the slab are equal to zero. That is, the surroundings ``pressure'' forces (buoyancy forces) are equal to gravity forces. The buoyancy forces are proportional to the ratio of the density of the slab to surrounding layer density. Thus, the stability question is whether the slab density from layer \(h\), \(\rho^{'}(h)\) undergoing a free expansion is higher or lower than the density of the layer \(h+dh\). If \(\rho^{'}(h) > \rho(h+dh)\) then the situation is stable. The term \(\rho^{'}(h)\) is slab from layer h that had undergone the free expansion. The reason that the free expansion is chosen to explain the process that the slab undergoes when it moves from layer \(h\) to layer \(h+dh\) is because it is the simplest. In reality, the free expansion is not far way from the actual process. The two processes that occurred here are thermal and the change of pressure (at the speed of sound). The thermal process is in the range of [cm/sec] while the speed of sound is about 300 [m/sec]. That is, the pressure process is about thousands times faster than the thermal process. The second issue that occurs during the "expansion'' is the shock (in the reverse case \([h+dh]\rightarrow h\)). However, this shock is insignificant (check book on Fundamentals of Compressible Flow Mechanics by this author on the French problem). The slab density at layer \(h+dh\) can be obtained using equation (101) as following
\[
\dfrac{\rho(h +dh) }{\rho(h)} = \dfrac{P\,T_0}{P_0\,T}
= {\left( 1 - \dfrac{C_x\, dh}{T_0} \right) ^
{\left( \dfrac{g }{R \,C_x} \right) } }
{\left( 1 + \dfrac{C_x\, dh}{T} \right)}
\label{static:eq:TxxRhodh}
\]
\[
\dfrac{\rho'(h+dh)}{\rho(h)} =
\left( \dfrac{P'(h+dh)}{P(h)} \right) ^{1/k}
\label{static:eq:dhInsentropic}
\]
When the symbol ′ denotes the slab that moves from layer \(h\) to layer \(h+dh\). The pressure ratio is given by equation (98) but can be approximated by equation (99) and thus
\[
\dfrac{\rho'(h+dh)}{\rho(h)} =
\left( 1 - \dfrac{g\, dh}{T(h) \;R } \right) ^{1/k}
\label{static:eq:dhInsentropicPtmp}
\]
Again using the ideal gas model for equation (105) transformed into
\[
\dfrac{\rho'(h+dh)}{\rho(h)} =
\left( 1 - \dfrac{\rho\, g dh}{P} \right) ^{1/k}
\label{static:eq:dhInsentropicP}
\]
Expanding equation (105) in Taylor series results in
\[
\left( 1 - \dfrac{\rho\, g dh}{P} \right) ^{1/k} =
1-\dfrac{g\,\rho\,dh}{P\,k}
-\dfrac{\left(g^2\,\rho^2\,k-g^2\,\rho^2\right)\,dh^2}
{2\,P^2\,k^2}
-...
\label{static:eq:dhInsentropicPExpansion}
\]
The density at layer h+dh can be obtained from (102) and then it is expanded in taylor series as
\[
\dfrac{\rho(h +dh) }{\rho(h)}
= {\left( 1 - \dfrac{C_x\, dh}{T_0} \right) ^
{\left( \dfrac{g }{R \,C_x} \right) } }
{\left( 1 + \dfrac{C_x\, dh}{T} \right)} \\
\sim 1 - \left( \dfrac{g\,\rho }{P} - \dfrac{C_x}{T}
\right) dh + \cdots \quad
\label{static:eq:dhTxxRho}
\]
The comparison of the right hand terms of equations (107) and (106) provides the conditions From a mathematical point of view, to keep the inequality for a small \(dh\) only the first term need to be compared as
\[
\dfrac{g\,\rho}{P\,k} >
\dfrac{g\,\rho }{P} - \dfrac{C_x}{T}
\label{static:eq:termoStability}
\]
After rearrangement of the inequality (108) and using the ideal gas identity, it transformed to
\[
\nonumber
\dfrac{C_x}{T} > \dfrac{(k-1)\,g\,\rho}{k\, P}\
C_x < \dfrac{k - 1 }{ k} \dfrac{g}{R}
\label{static:eq:thSbl}
\]
The analysis shows that the maximum amount depends on the gravity and gas properties. It should be noted that this value should be changed a bit since the k should be replaced by polytropic expansion \(n\). When lapse rate \(C_x\) is equal to the right hand side of the inequality, it is said that situation is neutral. However, one has to bear in mind that this analysis only provides a range and isn't exact. Thus, around this value additional analysis is needed . One of the common question this author has been asked is about the forces of continuation. What is the source of the force(s) that make this situation when unstable continue to be unstable? Supposed that the situation became unstable and the layers have been exchanged, would the situation become stable now? One has to remember that temperature gradient forces continuous heat transfer which the source temperature change after the movement to the new layer. Thus, the unstable situation is continuously unstable.
Contributors and Attributions
Dr. Genick Bar-Meir. Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or later or Potto license.


