3.1: Vapor Pressure Curve
- Page ID
- 7482
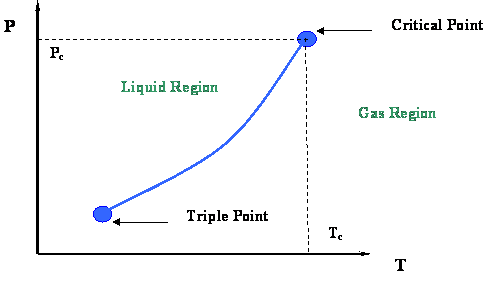
From the last module you will recall that the vapor-pressure curve (Figure 2.2.3) and its extremities were examined. That figure is presented in this module in Figure 3.1.1. We concluded that two very important thermodynamic points bound the vapor pressure curve: the Critical Point at its upper end and the Triple Point at its lower end.
Now let us take a second look at this figure. We can better understand the information represented by the vapor pressure curve by looking at the process of isobaric heating. This is illustrated by line ACB in Figure 3.1.2. The term “isobaric” pertains to a constant pressure process. By isobaric heating process, we then mean a “constant pressure addition of heat into a system.”
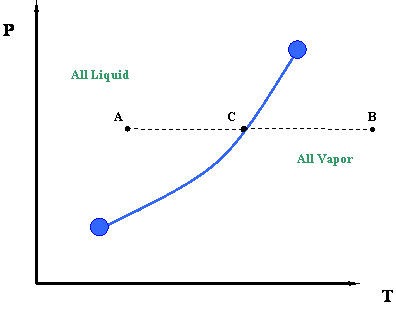
Such a process can be described as follows. Starting at point A and moving towards point C at constant pressure, we add heat to the system. By adding this heat, we cause a temperature increase in the system (Temperature at C > Temperature at A.). At point C, which lies above the vapor pressure or boiling point curve, we encounter a phase change. To the left of point C, at lower temperatures, exists only liquid. To the right of point C, at higher temperatures, there is only vapor. Therefore, a sharp discontinuity in density exists at point C. During this transition, from liquid to vapor, we will notice that the heat that we add to the system does not cause any temperature increase, and in fact, temperature and pressure conditions remain constant during the transition represented by the vapor curve. In other words, even though we are adding heat, the system remains at the pressure and temperature associated with point C until the whole phase transition has taken place — i.e., until all the vapor is converted to liquid. Instead of working to increase liquid temperature, this heat serves to move liquid molecules apart until all liquid has become vapor.
Up to this point, we saw that the heat added before the system reached the phase transition was used to raise the temperature of the substance. However, the heat that we are adding right now, during the phase transition, is not causing any temperature increase (hence it is said to be hidden heat or latent heat of vaporization). Therefore, we differentiate between two kinds of heat: sensible heat and latent heat.
- Sensible Heat: Its main purpose is to cause an increase in temperature of the system.
- Latent Heat: It serves only one purpose: to convert the liquid into vapor. It does not cause a temperature increase.
In fact, the name “latent” suggests “hidden.” Here, we are adding heat to the system but are not seeing its effect in terms of temperature increase. The heat that is needed to transform one mole of saturated liquid into vapor is known as the latent molar heat of vaporization: \( (\Delta \tilde{H}_{vap}) \)
Once we have converted all the liquid into vapor (i.e., we supplied all the necessary latent heat to accomplish this), we may continue to add more heat. If we do so, the temperature will rise again and we will end at point B (Figure 3.1.2). This heat is also sensible since it is causing the temperature of the system to rise.
It is interesting to note that, in order to reverse the process from point B to point A, we will have to remove the exact amount of heat that we had added before. This is a basic consequence of an energy balance principle. We call such a reverse process an isobaric cooling process. We will have to remove some sensible heat in order to cool the vapor from point B to C, and then we will remove all the latent heat of the vapor to condense it into liquid (transition at point C). Finally, we will also need to remove more sensible heat from the system for the cooling of the liquid from point C to point A.
In the previous two processes, from A to B or vice versa, we had to cross the phase boundary represented by the vapor pressure curve. However, this is not the only thermodynamic path that is available for us to go from A to B. Figure 3.1.3 depicts another possible path.
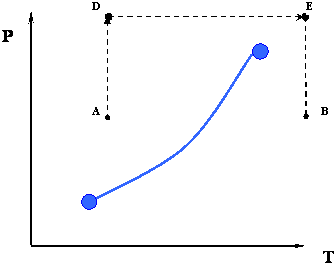
Instead of doing the whole process isobarically, we may devise a new path that may also accomplish the goal of taking the system from a condition ‘A’ to a condition ‘B.’ Consider the path ADEB that is shown in Figure 3.1.3.
Sequence of Paths
- Path AD: Isothermal compression
- Path DE: Isobaric heating
- Path EB: Isothermal expansion
There is something remarkable about this new path. Unlike the previous path, notice that we do not cross the phase boundary at all. The consequences of taking this new road may seem astonishing at first glance: we went from an all-liquid condition (point A) to an all-vapor condition (point B) without any sharp phase transition. In fact, along the path ADEB there is NO phase transition because we never crossed the phase boundary. Since the phase boundary represents a sharp discontinuity in density (and other physical properties), the fact that we are not crossing it tells us that as we go, there is actually a gradation in density (from a liquid like, or high-density at point A to a gas like, or low-density at point B) instead of a sharp change from the high liquid density to the low gas density.
We were able to do so because we went above the critical conditions. In the vicinity of and beyond critical conditions, we are no longer able to distinctly label the single-phase condition as either “liquid” or “gas”. Under these conditions, any transition takes place gradually without any differentiation between a ‘liquid’ and a ‘gas’ phase. We call this fluid, which we cannot define either as a liquid or as a gas, a supercritical fluid. In terms of density, a supercritical fluid may be described, at the same time, as a light liquid (its density is not as high as the liquid density) and a heavy gas (its density is not as low as the typical gas density of the given substance). The behavior of fluid around this area is an active and interesting area of current research.
In summary, for a pure substance, you can avoid having an abrupt phase transition (such as the one described by the path ACB) by going around the critical point (path ADEB). Keep in mind that any path that crosses the vapor pressure curve (ACB) will undergo a phase transition.


