27.3: Classes of Materials
- Page ID
- 32806
It can often be quite straightforward to tell materials classes apart by look or feel. Metals are usually more reflective or 'metallic' looking, ceramics are commonly matte and polymers may be shiny or matte, but are typically less dense than either metals or ceramics. Composites may be harder to immediately identify, but the surface may appear non-uniform and/or sectioning the sample may reveal fibres or particles.
It is useful to note that taking a cross-section can often be helpful in identifying materials used in components as the internal material and/or microstructure may differ from that at the edge.
Some materials may not be quite so easily identified simply from appearance and texture, but considering the factors mentioned can help to narrow down the possibilities. The table below gives a rough guideline to the kinds of properties you would expect from each class, and a few examples:
| Class | Common Properties | Examples |
|---|---|---|
| Metal | Hard, ductile and conduct heat and electricity | Copper (wires), stainless steel (cutlery) |
| Polymer | Widely variable, often soft and flexible | Polystyrene (cups), polycarbonate (CDs), polyethylene (plastic bags) |
| Ceramic | Hard, brittle, resistant to corrosion, electrically non-conductive | Concrete (buildings), PZT (piezoelectric used in lighters and ultrasonic transducers), porcelain (vases, teacups) |
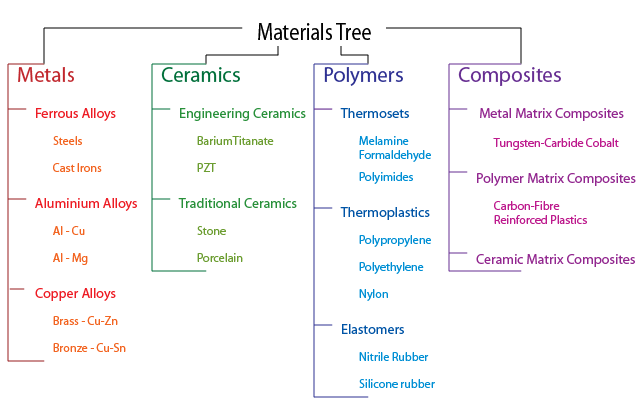
The tree-diagram below shows an overview of a variety of materials that might be encountered:
Coatings
Many components may have some kind of coating; a covering of another material designed to improve the surface qualities of the item. The improvement could be for many reasons including: corrosion resistance, appearance, adhesion, wear resistance and scratch resistance.
Different kinds of coating will have different processing methods. It is often possible to deduce the method from the composition (of the coating and the bulk of the component) and the shape of the component.
Common coating methods include:
Hot dip coating – a method used for coating metals (commonly ferrous alloys) with a low melting point alloy. The component is dipped in a bath of the molten coating alloy. For example zinc is often hot dipped onto steel (called ‘galvanising’). This also offers sacrificial corrosion protection and gives a distinctive ‘spangled’ appearance (which can be prevented by including particles to encourage nucleation in the electrolyte).

Steel coated with zinc
Electroplating – reduction of cations in an electrolytic solution onto conducting components. For example silver plated cutlery.
Anodising – commonly used for aluminium components, an electrochemical cell is set up which drives the oxidation of the metal, increasing the thickness of the protective oxide layer.
Vacuum deposition – also known as PVD – physical vapour deposition. For example, ‘evaporation’ involves the heating of the coating metal in a vacuum, so that it evaporates and is deposited onto the surface of the component that is positioned above. This process is used to make mirrors, depositing a thin layer of metal, usually aluminium.
Thermal Spraying - powder particles are fed into a high temperature torch (combustion or plasma), where they melt and are accelerated against the substrate. It is mainly used to produce relatively thick ceramic and metallic layers.
Enamelling - a powder is distributed on a surface, which is then heated so that the powder melts and bonds to the substrate. The resultant layer is usually glassy. Originally developed in ancient Egypt, and extensively used for jewelry, it is also widely employed for cooking utensils and various domestic items, especially those subjected to high temperature.
One example of a coated metal is shown below; a drawing pin, which appears to be brass, was found to be magnetic and so the surface was abraded, revealing a grey metal within – steel. Steel, which has very good mechanical properties, is covered with brass for aesthetic reasons and also protects the surface from corrosion.
 |
 |
| Magnetic drawing pin | Abraded pin revealing steel |


