32.4: Measurement of Tg
- Page ID
- 33015
There are several methods available to measure the glass transition temperature, some of which are given below. Since the value of the glass transition temperature depends on the strain rate and cooling or heating rate, there cannot be an exact value for Tg.
Mechanical Methods
It is possible to calculate a value for the glass transition temperature by measuring the elastic modulus of the polymer as a function of the temperature, for example by using a torsion pendulum. Around Tg there is a large fall in the value of the modulus. The frequency of the oscillation is important, since Tg depends on the time allowed for chain segment rotation.
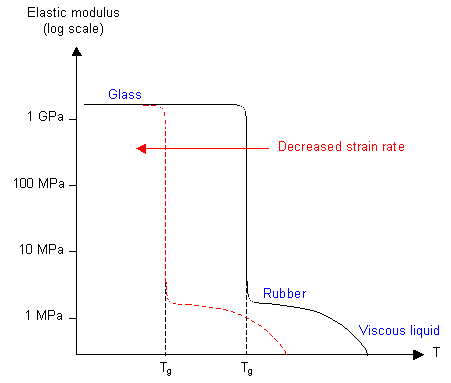
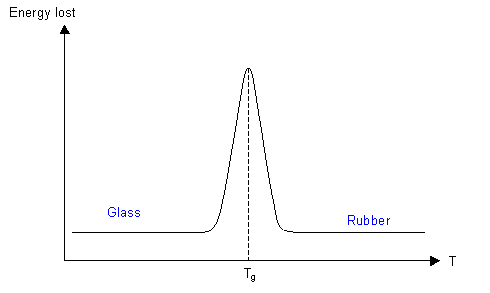
A more common method is dynamic mechanical thermal analysis (DMTA), which measures the energy absorbed when a specimen is deformed cyclically as a function of the temperature, and a plot of energy loss per cycle as a function of temperature shows a maximum at Tg.
Thermal Methods
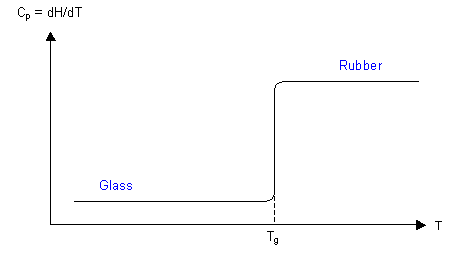
As was shown in the thermodynamic approach to glasses, the enthalpy of a polymer decreases as the temperature decreases, but with a change in slope in the graph at Tg. Taking the derivative of this graph with respect to temperature, the specific heat capacity can be plotted, as below. The specific heat capacity, Cp, can be measured using calorimetry, e.g. differential scanning calorimetry (DSC). The value of Tg depends on the heating or cooling rate.
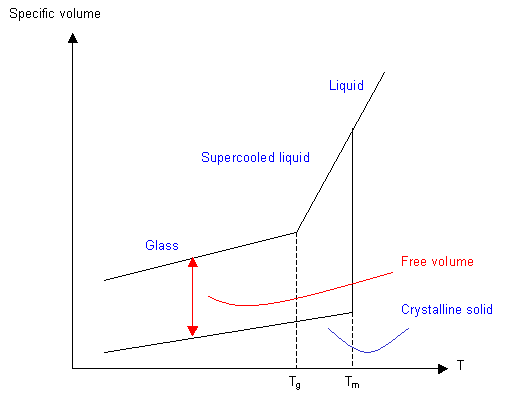
Volume Methods
The changes in conformation that occur above Tg require more volume, so plotting a graph of specific volume or thermal expansion coefficient against temperature will give a value for Tg. The actual volume of the molecules stays the same through Tg, but the free volume (the volume through which they can move) increases.
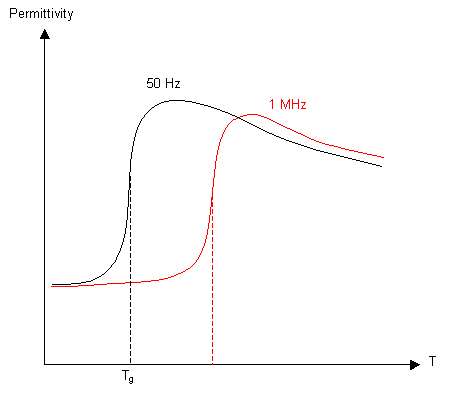
Dielectric Constant
If a varying electric field is applied to a polymeric material, any polar groups will align with the field. Below Tg rotation of the bonds is not possible, so the permittivity will be low, with a big increase around Tg. At higher temperatures the increased thermal vibrations cause the permittivity to drop again. If the frequency of the field is increased, the polar groups have less time to align, so the glass transition occurs at a higher temperature.