4.2: Retrograde Phenomenon
- Page ID
- 7490
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Regarding multi-component mixtures (where the binary system is the simplest case), some interesting phenomenon profoundly differentiate their behavior from the behavior of single-component systems. We are now talking about retrograde phenomena.
In the previous section, we learned how the “critical point” for a single-component system meant everything: the highest pressure and temperature at which two phases can exist and the point for which liquid and vapor phases are indistinguishable. Then we learned that this is not the case for multicomponent systems. Although the critical point for these systems is the common point between the dew and bubble point curves (the point for which liquid and vapor phases are indistinguishable), in general, this point neither represents the maximum pressure nor the maximum temperature for vapor-liquid coexistence. In fact, we gave new names to these maxima: cricondenbar (for the maximum pressure) and cricondentherm (for the maximum temperature). Let’s look at this again in Figure 4.2.5, where the critical point (Pc, Tc), cricondentherm (Tcc), and cricondenbar (Pcc) are highlighted.

Please recall that the bubble point curve represents the line of saturated liquid (100 % liquid with an infinitesimal amount of vapor) and the dew point curve represents the line of saturated vapor (100 % vapor with an infinitesimal amount of liquid). These conditions are all shown in Figure 4.2.5.
Let us now consider the isothermal processes taking place at T = T1 and T = T2, represented in Figure 4.2.6.
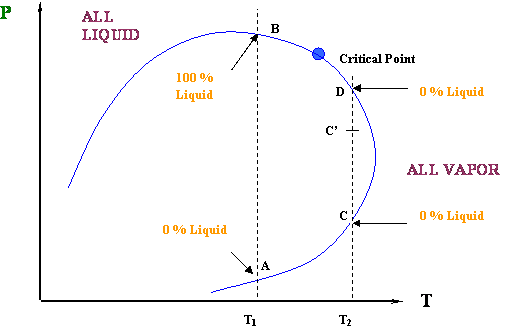
Figure 4.2.6 shows us two cases of isothermal compression for two different temperatures T1 and T2. Notice that these temperatures are such that T1 < Tc and Tc < T2 < Tcc.
It is common knowledge that an isothermal compression (increasing pressure while temperature is held constant) causes the condensation of a vapor (steam, in the case of water). That is the normal or expected behavior of a vapor under compression: the more you compress it, the more liquid you get out of it after the saturation conditions have been reached. This is always true for a pure-component system, such as water.
Well, that is exactly what is happening for our first case, the isothermal compression at T = T1. At point A, we are in an ALL VAPOR condition (0 % liquid) and we are starting to cross over into the two-phase region. As we compress from point A to B, more and more liquid is formed until the entire system has been condensed (point B). We went all the way from 0 % liquid to 100 % liquid, as we expected, by compressing the vapor. How liquid yield progresses with pressure is shown in Figure 4.2.7.

Again, there is nothing contrary to expectations here, and we would get the same result as long as T < Tc. However, there is something very interesting going on within the region Tc < T < Tcc.
In the second case (Tc < T2 < Tcc), we have a different behavior. At point C (Figure 4.2.6), we are starting in an ALL VAPOR condition (0 % liquid); by increasing pressure, we force the system to enter the two-phase region. Thus, some liquid has to drop out; we expect that as the pressure keeps increasing, we will produce more and more liquid. That is true to some extent… BUT, look at the final point of our journey, point D: although we are producing liquid, our final condition (dew point) requires us to have 0 % liquid in the system again.
How so?? This is telling us that, as we are entering the two-phase region, we will start to produce some liquid; but, there will be a point (of maximum liquid yield) where that liquid will start to vaporize (point C’). In other words, even though we are compressing the system, liquid will vaporize and not condense. Isn’t this contrary to expectations? Yes, and that is why we call this a retrograde (contrary to expectation) behavior.
Figure 4.2.8 shows a typical curve for the variation of the liquid volume percentage with pressure. This curve can be also referred to as the liquid dropout curve.

The increase in the liquid fraction with decreasing pressure between points C and D is exactly the opposite of the normal trend. This behavior, however, is typical of gas condensate systems. Retrograde conditions may be encountered in deep-well gas production, as well as in reservoir conditions.
For production operations, usually the objective is to maintain pressure so as to achieve maximum liquid dropout. The initial PVT conditions of the well may correspond to a point above point D. If the conditions at the wellhead are then maintained near point C’, liquid recovery is maximized at the surface. However, maximum liquid dropout is not always sought. At reservoir conditions, presence of liquid is not desirable in a gas reservoir, because liquids have negligible mobility (at low saturations) and thus, the hydrocarbon would be — for practical purposes — lost forever. Liquid also impairs gas mobility; hence, liquid production at reservoir conditions is to be avoided at all times in a gas reservoir. This is often achieved by repressurization or lean gas injection.
It is also important to see that a similar behavior is to be expected within the region Pc < P < Pcc. In this case, we talk about retrograde vaporization since we will be moving from a 100 % liquid to another 100 % liquid condition (both on the bubble point curve) in an isobaric heating.


