3.1: Four Questions
- Page ID
- 81483
When developing an accounting or balance equation for a new property, there are four questions that must be answered:
- What is it?
- How can it be stored in a system?
- How can it be transported across the boundary of a system?
- How can it be generated or consumed?
In this section, we will use these questions to guide us as we develop a mass balance built on a fundamental physical principle the conservation of mass.
3.1.1 What is mass?
Mass can be described in many different ways. Newton described it as a "measure of the inertia" of a body; however, this is not very useful without defining inertia. Similarly, we could define it as the amount of a substance; however, this is also faulty precisely because it neglects the inertia idea.
This problem of definition is a common one in science and language in general. Any definition must of necessity build upon other words that are assumed to be understood. In science and engineering, we will make use of an operational definition to provide a precise definition of a new term or concept. An operational definition is a series of steps or operations that must be performed to define the quantity or concept in question.
Using this approach, the mass of any object could be described in terms of the mass of a standard or reference object:
The ratio of the gravitational mass of an object to the gravitational mass of a standard or reference object equals the ratio of the weight of the object of unknown mass to the weight of the standard object.
This definition assumes that the concept of weight is understood. An alternative definition could be developed from Newton's second law that relates the force \(\vec{F}\), the system mass \(m\) and acceleration \(\vec{a}\), \(\vec{F} = m \vec{a}\):
The ratio of the inertial mass of an object to the inertial mass of standard or reference object equals the ratio of the acceleration of the object to the acceleration of the reference object when both objects are subjected to the same force.
As you have learned in physics, the inertial mass and the gravitational mass of an object are equal. For our purposes, we will take the definition of mass as being an undefined term and rely on your background from physics.
3.1.2 How can mass be stored in a system?
Mass is an intrinsic property of matter. Any system that contains matter has mass. If the mass of a particle is \(m_i\), then the mass of a system of particles \(m_{sys}\) is equal to the sum of the mass of the individual particles:
\[ m_{sys} = \sum_{i=1}^n m_i \nonumber \]
Since the mass of a system depends on the number of particles (or extent) of the system, we recognize that mass is also an extensive property.
More generally, the mass of a system can be found by integrating the mass density \(\rho\) over the volume of the system \(V_{sys}\).
\[ m_{sys} = \int \limits_{V_{sys}} \rho \, dV \nonumber \]
Mass density or more commonly just the density of a substance is defined as the mass per unit volume. Because it has a value at a point, density is an intensive property. The dimensions of density are \([\text{M}]/[\text{L}]^3\). Typical units for density are \(\text{kg/m}^3\) in SI and \(\text{lb} \cdot \text{m/ft}^3\) in USCS.
In general, substances can be classified as either incompressible or compressible. An incompressible substance is one whose density is constant with regard to both space and time. There are no truly incompressible substances; however, many substances may be modeled as incompressible under certain conditions. For example, the hydraulic fluid in your automobile brake lines is essentially incompressible. The same is true for most liquids and solids. A compressible substance is one whose density can change significantly during a change in state. Gases and vapors fall in this category.
.png?revision=1)
Figure \(\PageIndex{1}\): System
To better understand the significance of this simple relation, consider the system and coordinate system in Figure 3-1. Depending upon whether it is an open or closed system, the mass of a system may change with time. Mathematically, we would say that the mass of the system depends upon time, \(m_{sys} = m_{sys}(t)\). The mass density \(\rho\) at any point in the system may depend on both its position \((x,y,z)\) and time \((t)\), i.e. \(\rho = \rho (x,y,z,t)\). Substituting these terms back into Eq. \(\PageIndex{2}\) we have
\[ m_{sys} (t) = \int\limits_{V_{sys}} \rho (x,y,z,t) \, dV \nonumber \]
Notice how the integration over the system volume removes the spatial dependence leaving only the time dependence. Typically we will not write out the spatial and time dependence as in Eq. \(\PageIndex{3}\); however, you should remember that these variations may exist.
A solar pond contains a stratified mixture of salt and water. The solar pond has a rectangular section surface area of \(1000 \ \text{m}^2\) and a uniform depth of \(6 \ \text{m}\). The density of the salt water mixture varies with the distance below the surface \(h\) according to the relation \(\rho / \rho_{H2O} = 1+0.001(h/\text{m})^2\) where \(\rho_{H2O} = 998 \text{kg/m}^3\). Determine the mass of salt water in the pond, in kilograms.
Solution
Known: A solar pond contains a stratified mixture of salt and water.
Find: The mass of salt water in the pond, in \(\text{kg}\).
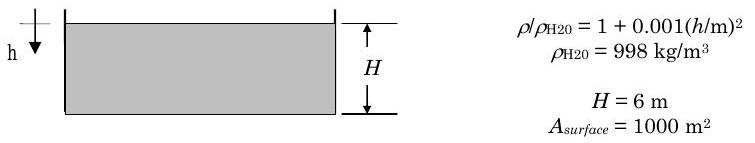
Analysis:
We start with the mathematical relation that defines the mass inside a system in terms of its mass density and volume:
\[ m_{sys(t)} = \int\limits_{V_{sys}} \rho _{(x, y, z, t)} \, dV \nonumber \]
Because the density only depends on the depth \(h\), it makes sense to define the differential volume as \(dV = A_{surface} \ dh\) and the limits of integration from \(0 \to H\). Combining this with the density relationship gives
\[ \begin{align*} m_{sys} &= \int\limits_{0}^{H} \rho_{H2O} \left[ 1 + 0.001 \left( \frac{h}{\text{m}} \right) ^2 \right] \underbrace{ \left( A_{surface} \, dh \right) }_{V_{sys}} \\ &= \rho_{H2O} A_{surface} \left. \left[ h + \frac{0.001}{3} \frac{h^3}{\text{m} ^2} \right] \ \right\vert_{0}^{H} \\ &= \rho_{H2O} A_{surface} \left[ H + \frac{0.001}{3} \frac{H^3}{\text{m} ^2} \right] = \underbrace{ \rho_{H2O} \left[ 1 + \frac{0.001}{3} \left( \frac{H}{\text{m}} \right)^2 \right] }_{\rho_{average}} \underbrace{ \left( A_{surface} H \right) }_{V_{sys}} \end{align*} \nonumber \]
Substituting in the numbers, we have
\[ \begin{align*} m_{pond} &= \left( 998 \frac{ \text{kg} }{ \text{m}^3 } \right) \times \left[ 1 + \frac{0.001}{3} \left( \frac{6 \ \text{m}}{\text{m}} \right)^2 \right] \times \left[ \left( 1000 \ \text{m}^2 \right) \left( 6 \ \text{m} \right) \right] \\ &= \left[ (998) (1 + 0.012) (6000) \right] \left[ \frac{ \text{kg} }{ \text{m}^3 } \cdot \text{m}^2 \cdot \text{m} \right] \\ &= 6.06 \times 10^6 \ \text{kg} \end{align*} \nonumber \]
Comments:
- Notice the form of the equation for the density. This equation is dimensionally homogeneous because it works correctly with any set of units. If the equation had been presented without the meter unit in the expression, i.e. \(\rho / \rho_{H2O} = 1+0.001(h)^2\) instead of \(\rho / \rho_{H2O} = 1+0.001(h/ \text{m})^2\), the equation could have only been used correctly if numerical values of \(h\) were always supplied in meters.
- Also notice how the calculations were first done symbolically. This is the preferred method for solving problems, because it allows you to keep track of the physics without getting lost in the numbers. Notice how we were able to identify the volume of the pond, \(V_{pond}\), and the average density, \(\rho_{avg}\), in the last step of the calculation. Constantly connecting our mathematics with our physical understanding of the problem provides a continuous check on our work.
- When substituting in the magnitude of the physical quantities, \(H\( and \(\rho_{H2O}\), notice how we substitute in both a number and a unit. Strictly speaking, if we were to leave off the units the expression would be mathematically incorrect. One approach to simplify the calculations, especially when you are dealing with unfamiliar units, is to segregate the numerical and the unit calculations into two parts as shown in the problem.
3.1.3 How can mass be transported across a system boundary?
There are two ways that mass can cross the boundary of a system:
- Gross motion of mass across the boundary.
- Microscopic motion of mass due to molecular diffusion.
In this course we will focus on only the first mechanism. Specifically we will define the mass flow rate \(\dot{m}\) as the time rate at which mass crosses a boundary. The mass flow rate has dimensions of \([\text{M}]/[\text{T}]\). Typical units are \(\text{kg/s}\) in SI, and \(\text{lb m/s}\) or \(\text{slugs/s}\) in USCS.
3.1.4 How can mass be created or destroyed?
Based on your past experiences, how would you answer this question? If you said, "It can't!" you are recognizing a fundamental physical law known as the Conservation of Mass. Recognizing that mass is a property, we can state this law as follows: Mass is a conserved property.
Specifically this means that, excluding energy-to-mass conversion, it is impossible to generate or consume mass.
3.1.5 Putting it all together
Combining what we have learned from the first four sections about mass, we can use our accounting or balance framework to write the following conservation of mass equation or mass balance:
\[ \frac{d m_{sys}}{dt} = \sum_{in} \dot{m}_i - \sum_{out} \dot{m}_e \nonumber \]
where the first summation is over all the inlets (entrances) and the second summation is over all of the outlets (exits). In words, we might say that
The time rate-of-change of the mass in the system equals the sum of the mass flow rates into system minus the sum of the mass flow rates out of the system.
Another statement might be
The rate of accumulation of mass within the system equals the net mass flow rate into the system.
As with any of our balance equations, please note that all of the terms in the balance equation have independent definitions and can be calculated independently of the other expressions. The unique contribution of this mass balance and its related physical law, The Conservation of Mass, is that we now have a unique relationship between all of these quantities.


