10.2: Sample Preparation
- Page ID
- 8228
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)For Metals
When preparing samples for microscopy, it is important to produce something that is representative of the whole specimen. It is not always possible to achieve this with a single sample. Indeed, it is always good practice to mount samples from a material under study in more than one orientation. The variation in material properties will affect how the preparation should be handled, for example very soft or ductile materials may be difficult to polish mechanically.
Cutting a specimen
It important to be alert to the fact that preparation of a specimen may change the microstructure of the material, for example through heating, chemical attack, or mechanical damage. The amount of damage depends on the method by which the specimen is cut and the material itself.
Cutting with abrasives may cause a large amount of damage, whilst the use of a low-speed diamond saw can cause fewer problems. There are many different cutting methods, although some are used only for specific specimen types.
Mounting
Mounting of specimens is usually necessary to allow them to be handled easily. It also minimises the amount of damage likely to be caused to the specimen itself.
The mounting material used should not influence the specimen as a result of chemical reaction or mechanical stresses. It should adhere well to the specimen and, if the specimen is to be electropolished (an Electrolytic process ) or examined under a Scanning Electron Microscope , then the mounting material should also be electrically conducting.
Specimens can be hot mounted (at around 200 °C) using a mounting press, either in a thermosetting plastic (e.g. phenolic resin), or a thermosoftening plastic (e.g. acrylic resin). If hot mounting will alter the structure of the specimen a cold-setting resin can be used, e.g. epoxy, acrylic or polyester resin. Porous materials must be impregnated by resin before mounting or polishing, to prevent grit, polishing media or etchant being trapped in the pores, and to preserve the open structure of the material.
A mounted specimen usually has a thickness of about half its diameter, to prevent rocking during grinding and polishing. The edges of the mounted specimen should be rounded to minimise the damage to grinding and polishing discs.
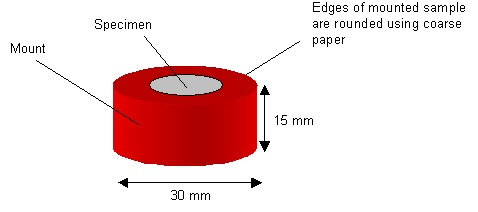
A diagram of a mounted specimen
Grinding
Surface layers damaged by cutting must be removed by grinding. Mounted specimens are ground with rotating discs of abrasive paper flushed with a suitable coolant to remove debris and heat, for example wet silicon carbide paper. The coarseness of the paper is indicated by a number: the number of grains of silicon carbide per square inch. So, for example, 180 grit paper is coarser than 1200.
The grinding procedure involves several stages, using a finer paper (higher number) for each successive stage. Each grinding stage removes the scratches from the previous coarser paper. This is more easily achieved by orienting the specimen perpendicular to the previous scratches, and watching for these previously oriented scratches to be obliterated. Between each grade the specimen is washed thoroughly with soapy water to prevent contamination from coarser grit present on the specimen surface. Typically, the finest grade of paper used is the 1200, and once the only scratches left on the specimen are from this grade, the specimen is thoroughly washed with water, followed by alcohol and then allowed to dry.
It is possible to determine the start point for grinding using the following empirical relationship where the width of the largest scratch is measured under a microscope:
![]()
This prevents putting more damage into the sample than already exists; the coarsest grades of paper are often not useful.
Cleaning specimens in an ultrasonic bath can also be helpful, but is not essential.
The series of photos below shows the progression of the specimen when ground with progressively finer paper.
|
|
|
| Copper specimen ground with 180 grit paper | Copper specimen ground with 400 grit paper |
|
|
|
| Copper specimen ground with 800 grit paper | Copper specimen ground with 1200 grit paper |
Polishing
Polishing discs are covered with soft cloth impregnated with abrasive diamond particles and an oily lubricant. Particles of two different grades are used : a coarser polish - typically with diamond particles 6 microns in diameter which should remove the scratches produced from the finest grinding stage, and a finer polish – typically with diamond particles 1 micron in diameter, to produce a smooth surface. Before using a finer polishing wheel the specimen should be washed thoroughly with warm soapy water followed by alcohol to prevent contamination of the disc.
|
|
|
|
Copper specimen polished to 6 micron level |
Copper specimen polished to 1 micron level. Ideally there should be no scatches after polishing, but it is often hard to completely remove them all. |
Mechanical polishing will always leave a layer of disturbed material on the surface of the specimen, if the specimen is particularly susceptible to mechanical damage (or excessive force is used in the grinding and polishing stages) debris can become embedded in the surface and plastic deformation may exist below the surface. Electropolishing or chemical polishing can be used to remove this, leaving an undisturbed surface.
Etching
Etching is used to reveal the microstructure of the metal through selective chemical attack. It also removes the thin, highly deformed layer introduced during grinding and polishing.
In alloys with more than one phase, etching creates contrast between different regions through differences in topography or reflectivity. The rate of etching is affected by crystallographic orientation, the phase present and the stability of the region. This means contrast may arise through different mechanisms – therefore revealing different features of the sample.
In all samples, etchants will preferentially attack high energy sites, such as boundaries and defects.
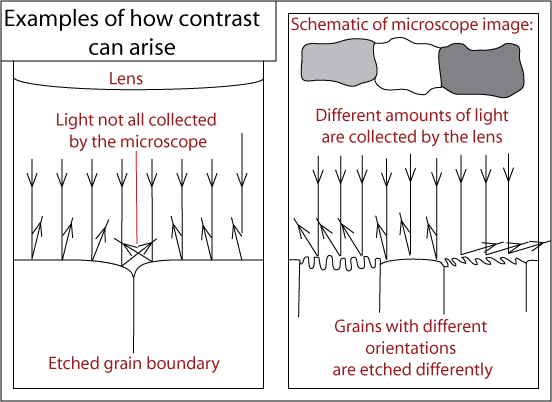
The specimen is etched using a reagent. For example, for etching stainless steel or copper and its alloys, a saturated aqueous solution of ferric chloride, containing a few drops of hydrochloric acid is used. This is applied using a cotton bud wiped over the surface a few times (Care should be taken not to over-etch - this is difficult to determine, however, the photos below may be of some help). The specimen should then immediately be washed in alcohol and dried.
Following the etching process there may be numerous small pits present on the surface. These are etch pits caused by localised chemical attack and, in most cases, they do not represent features of the microstructure. They may occur preferentially in regions of high local disorder, for example where there is a high concentrationof dislocations.
If the specimen is over etched, ie. etched for too long, these pits tend to grow, and obscure the main features to be observed. If this occurs it may be better to grind away the poorly etched surface and re-polish and etch, although it is important to remember what features you are trying to observe – repeatedly grinding a very thin sample may leave nothing to see.
|
|
|
|
Etched copper specimen |
Over etched copper specimen |
Ideally the surface to be examined optically should be flat and level. If it is not, the image will pass in and out of focus as the viewing area is moved across the surface. In addition, it will make it difficult to have the whole of the field of view in focus - while the centre is focused, the sides will be out of focus. By using a specimen levelling press (shown below) this problem can be avoided, as it presses the mounted specimen into plasticene on a microscope slide, making it level. A small piece of paper or cloth covers the surface of the specimen to avoid scratching.

Specimen levelling press










