Polymer Chemistry: The Glass Transition
- Page ID
- 2924
Semi-crystalline solids have both amorphous and crystalline regions. According to the temperature, the amorphous regions can be either in the glassy or rubbery state. The temperature at which the transition in the amorphous regions between the glassy and rubbery state occurs is called the glass transition temperature.
The Glass Transition
The glass transition is a property of only the amorphous portion of a semi-crystalline solid. The crystalline portion remains crystalline during the glass transition. At a low temperature the amorphous regions of a polymer are in the glassy state. In this state the molecules are frozen on place. They may be able to vibrate slightly, but do not have any segmental motion in which portions of the molecule wiggle around. In the glassy state, the motion of the red molecule in the schematic diagram below would NOT occur. When the amorphous regions of a polymer are in the glassy state, it generally will be hard, rigid, and brittle.
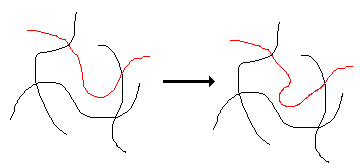
If the polymer is heated it eventually will reach its glass transition temperature. At this temperature portions of the molecules can start to wiggle around as is illustrated by the red molecule in the diagram above. The polymer now is in its rubbery state. The rubbery state lends softness and flexibility to a polymer.
You may have experienced the glass transition of chewing gum. At body temperature the gum is soft and pliable, which is characteristic of an amorphous solid in the rubbery state. If you put a cold drink in your mouth or hold an ice cube on the gum, it becomes hard and rigid. The glass transition temperature of the gum is somewhere between 0 °C and 37 °C.
The glass transition is NOT the same as melting.
Glass Transition
- Property of the amorphous region
- Below Tg: Disordered amorphous solid with immobile molecules
- Above Tg: Disordered amorphous solid in which portions of molecules can wiggle around
- A second order transition (see below)
Melting
- Property of the crystalline region
- Below Tm: Ordered crystalline solid
- Above Tm: Disordered melt
- A first-order transition (see below)
Thermodynamic transitions are classified as being first- or second-order. In a first-order transition there is a transfer of heat between system and surroundings and the system undergoes an abrupt volume change. In a second-order transition, there is no transfer of heat, but the heat capacity does change. The volume changes to accommodate the increased motion of the wiggling chains, but it does not change discontinuously. Illustrative plots of specific volume vs. temperature are shown at the right for amorphous and crystalline polymers.
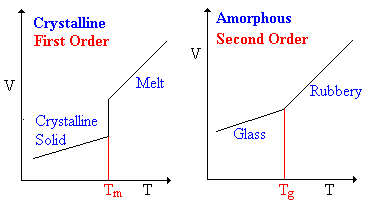
Glass Transition Temperature
When an amorphous polymer is heated, the temperature at which it changes from a glass to the rubbery form is called the glass transition temperature, Tg. A given polymer sample does not have a unique value of Tg because the glass phase is not at equilibrium. The measured value of Tg will depend on the molecular weight of the polymer, on its thermal history and age, on the measurement method, and on the rate of heating or cooling. Approximate glass transition temperatures of a few polymers are shown below.
| Polymer | Tg (°C) |
|---|---|
| Polyethylene (LDPE) | -125 |
| Polypropylene (atactic) | -20 |
| Poly(vinyl acetate) (PVAc) | 28 |
| Poly(ethyleneterephthalate) (PET) | 69 |
| Poly(vinyl alcohol) (PVA) | 85 |
| Poly(vinyl chloride) (PVC) | 81 |
| Polypropylene (isotactic) | 100 |
| Polystyrene | 100 |
| Poly(methylmethacrylate) (atactic) | 105 |
Contributors and Attributions
David Whisnant (Wofford College). Partial support for this work was provided by the National Science Foundation's Division of Undergraduate Education through grants DUE #9950809 and DUE #9950296. Additional support was provided by the Camille and Henry Dreyfus Foundation.


