7.4: Substance Models
- Page ID
- 81508
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In applying the conservation of energy equation to model a physical device, we must frequently evaluate changes in internal energy or enthalpy (en - thal ’- py). These in turn are related to the directly measurable properties such as pressure, temperature, and specific volume or density. The study of how thermophysical properties are related is one of the major aims of the study of thermodynamics.
For our present study we will consider only two substance models. These models will be presented without significant development or explanation. This will be covered in a later course. Each substance model provides us with an equation of state for an important class of substances. The major results of both models are summarized in Section 7.4.3, and you are encouraged to skip ahead and get an overall picture before you begin the more detailed descriptions of each model.
7.4.1 Ideal Gas Model with Room-Temperature Specific Heats
The ideal gas model is familiar to most students from physics and chemistry; however, we will use it more extensively than most of you have done previously. The ideal gas model is just that — a set of constitutive equations that accurately models the behavior of gases and vapors under certain restricted conditions.
Basic Assumptions
The ideal gas model is built upon three assumptions:
- Pressure, volume and temperature obey the ideal gas equation: \[P V=N R_{u} T=m R T \quad \text { where } R=\frac{R_{u}}{M} \nonumber \]
- The specific internal energy only depends on temperature: \[u=u(T) \nonumber \]
- The molar mass of an ideal gas is identical with the actual molar mass of the substance: \[M_{\text {ideal gas }}=M_{\text {real stuff }} \nonumber \] In addition, we will add one additional assumption that will greatly simplify our introduction to using ideal gas properties.
- The specific heats are independent of temperature. (In general, the specific heats do in fact change with temperature, and this variation will be considered later to increase the accuracy of the ideal gas model.) However, as a first approximation and a pretty accurate assumption for small temperature changes near room temperature, we will assume that the specific heats are constant and equal to the values at room temperature.
We'll refer to our ideal gas model that is based on all four assumptions as the "ideal gas model with room-temperature specific heats."
\(P \text{-} \upsilon \text{-} T\) Relationship
An ideal gas by definition satisfies the ideal gas equation, Eq. \(\PageIndex{1}\). Typically we find it most useful to rearrange this equation so it only includes intensive properties: \[P=\rho R T \quad \text { or } \quad P \upsilon = R T \nonumber \] where \(R\) is the specific gas constant \(\left(=R_{u} / M\right)\) with units of \(\mathrm{kJ} /(\mathrm{kg} \cdot \mathrm{K})\) or \((\mathrm{ft} \cdot \mathrm{lbf}) /\left(\mathrm{lbm} \cdot{ }^{\circ} \mathrm{R}\right)\). This equation was discussed extensively in Chapter 3 and you are encouraged to review this material.
Changes in \(u\) and \(h\)
The differential change in specific internal energy \(u\) for an ideal gas is calculated using the relation: \[d u=c_{v} \ d T \quad \text { [Ideal gas] } \nonumber \] where \(c_{v}\) is called "see-sub-vee" or the specific heat at constant volume (see note). To calculate the finite change in \(u\) for an ideal gas with constant specific heats, we integrate Eq. \(\PageIndex{5}\) between state 1 and state 2 and obtain the equation: \[\Delta u=u_{2}-u_{1}=c_{v}\left(T_{2}-T_{1}\right) \quad \left[\begin{array}{c} \text { Ideal gas, } \\ \text { constant } c_{v} \end{array}\right] \nonumber \] This equation can be used to calculate \(\Delta u\) for any process. It is not restricted to constant-volume processes.
The differential change in specific enthalpy \(h\) for an ideal gas is calculated using the relation: \[d h=c_{p} \ dT \quad \text { [Ideal gas] } \nonumber \] where \(c_{p}\) is called "see-sub-pee" or the specific heat at constant pressure (see note). To calculate the finite change in \(h\) for an ideal gas with constant specific heats, we integrate Eq. \(\PageIndex{7}\) between state 1 and state 2 and obtain the equation: \[\Delta h=h_{2}-h_{1}=c_{P}\left(T_{2}-T_{1}\right) \quad\left[\begin{array}{c} \text { Ideal gas, } \\ \text { constant } c_{p} \end{array}\right] \nonumber \] This equation can be used to calculate \(\Delta h\) for any process. It is not restricted to constant-pressure processes.
The specific heats \(c_{P}\) and \(c_{v}\) are defined mathematically as the following partial derivatives: \[c_{v} \equiv \left(\frac{\partial u}{\partial T}\right)_{v} \quad \text { and } \quad c_{P} \equiv \left(\frac{\partial h}{\partial T}\right)_{P} \nonumber \] where the phrase "constant volume" or "constant pressure" refers specifically to what is held constant during the differentiation.
Specific heat relationships
The specific heats for an ideal gas are related to the specific gas constant by the following equation: \[c_{P}-c_{v}=R \quad \text { [Ideal gas] } \nonumber \] Sometimes all we know (or remember for a gas is its specific heat ratio. The specific heat ratio is defined as \[k \equiv \frac{c_{P}}{c_{v}} \nonumber \]
Combining Eqs. \(\PageIndex{9}\) and \(\PageIndex{10}\) we have the following equations that relate the specific heats, the specific gas constant, and the ratio of specific heats. \[c_{v}=\frac{1}{k-1} R \quad \text { and } \quad c_{P}=\frac{k}{k-1} R \quad \text { [Ideal gas] } \nonumber \] These relations are frequently useful because the ratio of specific heats is a strong function of the molecular structure of the gas, i.e. whether it is monatomic, etc., and a weaker function of its temperature.
7.4.2 Incompressible Substance Model with Room-Temperature Specific Heats
The incompressible substance model is not as familiar as the ideal gas model. This model is based on observations of how liquids and solids behave. We will assume that this is an accurate model for most liquids and solids. It does not incorporate energy changes due to elastic strain of solids.
Basic Assumptions
The incompressible substance model is built upon three assumptions:
- Specific volume of an incompressible substance is a constant: \[v=\text { constant } \nonumber \] Values of specific volume will be evaluated at room temperature.
- The specific internal energy only depends on temperature: \[u=u(T) \nonumber \]
- The incompressible substance molar mass for a substance is identical with the actual molar mass of the substance: \[M_{\text {incompressible substance}}=M_{\text {real stuff }} \nonumber \] In addition, we will add one additional assumption that will greatly simplify our introduction to the incompressible substance model.
- The specific heats are independent of temperature. (In general, the specific heats do in fact change with temperature; however, the changes with temperature for most solids and liquids are small and this is an accurate assumption.) We will assume that the specific heats are constant and equal to the values at room temperature.
We'll refer to our incompressible substance model that is based on all four assumptions as the "incompressible substance model with room-temperature specific heats."
\(P \text{-} \upsilon \text{-} T\) Relationships
For an incompressible substance there is no relationship between the pressure, the specific volume, and the temperature of the substance. The value of the specific heat depends on the substance.
The densities of solids and liquids do in fact change slightly with temperature. Anyone who has overfilled an ice cube tray or dealt with frozen pipe understands this effect. For our purposes, we will assume that the value of the specific volume (or density) can be evaluated at room temperature conditions.
Changes in \(u\) and \(h\)
The differential change in specific internal energy \(u\) for an incompressible substance is calculated using the relation: \[d u=c_{v} \ dT \quad \text { [Incompressible substance] } \nonumber \] where \(c_{v}\) is called "see-sub-vee" or the specific heat at constant volume (See note above). To calculate the finite change in \(u\) for an incompressible substance with constant specific heats, we integrate Eq. \(\PageIndex{15}\) between state 1 and state 2 and obtain the equation: \[\Delta u = u_{2}-u_{1} = c_{v}\left(T_{2}-T_{1}\right) \quad\left[\begin{array}{c} \text { Incompressible substance, } \\ \text {constant } c_{v} \end{array}\right] \nonumber \] This equation can be used to calculate \(\Delta u\) for any process. It is not restricted to constant volume processes.
The differential change in specific enthalpy \(h\) for an incompressible substance is calculated using the relation: \[dh = d(u+P \upsilon) = du + \upsilon \ dP \quad \text { [Incompressible substance] } \nonumber \] where \(c_{p}\) is called "see-sub-pee" or the specific heat at constant pressure. To calculate the finite change in \(h\) for an incompressible substance with constant specific heats, we integrate Eq. \(\PageIndex{17}\) between state 1 and state 2 and obtain the equation: \[\begin{array}{l} \Delta h &= u_{2}-u_{1} + \upsilon\left(P_{2}-P_{1}\right) \\ &= c_{v}\left(T_{2}-T_{1}\right) + \upsilon\left(P_{2}-P_{1}\right) \quad\left[\begin{array}{c} \text { Incompressible substance, } \\ \text { constant } c_{v} \end{array}\right] \end{array} \nonumber \] This equation can be used to calculate \(\Delta h\) for any process.
Specific heat relationships
Using the basic assumptions for the incompressible substance, it can be shown that two specific heat at constant pressure and constant volume are equal: \[c_{P}=c_{v}=c \quad \text { [Incompressible substance] } \nonumber \] where \(c\) is sometimes just called the specific heat. Typically, tables of data for solids and liquids will only list values for \(c_{p}\) because it is the easiest to measure.
7.4.3 Summary of our substance models
The basic assumptions and key equations for each of our substance models are summarized in the following table. Once you have selected a substance model to apply in a given problem, this table gives all of the necessary equations for applying the model. Room-temperature values for the thermophysical properties for several gases, liquids, and solids are given in next two tables. One table gives values in SI and one gives values in USCS.
Substance models are required to relate properties in the energy balance like \(h\) and \(u\) to properties that are easy to measure like pressure, temperature, and specific volume.
| Two Substance Models (Constitutive Relations) | ||
|---|---|---|
| Equation of State | ||
| Ideal Gas Model with room-temperature specific heats |
Incompressible Substance Model with room-temperature specific heats |
|
| Used to model behavior of | gases and vapors | liquids and solids |
| Basic Model Assumptions |
|
|
| \(P \text{-} T \text{-} \rho\) and \(P \text{-} T \text{-} \upsilon\) relations | \(P = \rho RT\) and \(P \upsilon = RT\) where \(R=R_{u} / M\) |
\(\upsilon = 1 / \rho = \text{constant}\) Evaluated at room temperature |
| Specific heat relations | \(c_{P} = c_{v} = R; \quad k = c_{P} / c_{v}\) | \(c_{p} = c_{v} = c, \text{ a constant}\) |
| \(c_{P}\) and \(c_{V}\) values | Evaluated at room temperature | Evaluated at room temperature |
| \(\Delta u\) — specific internal energy | \(\Delta u = u_{2}-u_{1} = c_{v}\left(T_{2}-T_{1}\right)\) | \(\Delta u = u_{2}-u_{1} = c\left(T_{2}-T_{1}\right)\) |
| \(\Delta h\) — specific enthalpy | \(\Delta h = h_{2}-h_{1} = c_{P}\left(T_{2}-T_{1}\right)\) | \[ \begin{aligned} \Delta h &= h_{2}-h_{1} \\ &= \left(u_{2}+P_{2} \upsilon\right) - \left(u_{1}+P_{1} \upsilon\right) \\ &= \left(u_{2}-u_{1}\right) + \upsilon \left(P_{2}-P_{1}\right) \\[4pt] \text{thus } \Delta h &= \Delta u + \upsilon \Delta P = c \Delta T + \upsilon \Delta T \end{aligned} \nonumber \] |
|
\(\Delta s\) — specific entropy Note: All temperatures are absolute values, i.e. \(\mathrm{K}\) or \({ }^{\circ} \mathrm{R}\), in the entropy relations |
\[ \begin{aligned} \Delta s &= s_{2}-s_{1} \\ &= c_{P} \ln \left(T_{2}-T_{1}\right) - R \ln \left(P_{2}-P_{1}\right) \\ &= c_{v} \ln \left(T_{2}-T_{1}\right) + R \ln \left(\upsilon_{2}-\upsilon_{1}\right) \end{aligned} \nonumber \] | \[ \begin{aligned} \Delta s &= s_{2}-s_{1} \\ &= c \ln \left(T_{2}-T_{1}\right) \end{aligned} \nonumber \] |
| Gases (at \(25 ^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\)) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Substance | Molar Mass | \(\dfrac{R}{\left[ \dfrac{\mathrm{kJ}}{\mathrm{kg} \cdot \mathrm{K}}\right]}\) | \(\dfrac{c_{v}}{\left[ \dfrac{\mathrm{kJ}}{\mathrm{kg} \cdot \mathrm{K}}\right]}\) | \(\dfrac{c_{P}}{\left[ \dfrac{\mathrm{kJ}}{\mathrm{kg} \cdot \mathrm{K}}\right]}\) | \(k\) | \(\dfrac{T_{c}}{\mathrm{K}}\) | \(\dfrac{P_{c}}{\mathrm{bar}}\) | |
| Acetylene | \(\mathrm{C}_2 \mathrm{H}_2\) | \(26.04\) | \(0.3193\) | \(1.37\) | \(1.69\) | \(1.23\) | \(309\) | \(62.4\) |
| Air | --- | \(28.97\) | \(0.2870\) | \(0.718\) | \(1.005\) | \(1.40\) | \(133\) | \(37.7\) |
| Ammonia | \(\mathrm{NH}_3\) | \(17.04\) | \(0.4879\) | \(1.66\) | \(2.15\) | \(1.30\) | \(406\) | \(112.8\) |
| Carbon dioxide | \(\mathrm{CO}_2\) | \(44.01\) | \(0.1889\) | \(0.657\) | \(0.846\) | \(1.29\) | \(304.2\) | \(73.9\) |
| Carbon monoxide | \(\mathrm{CO}\) | \(28.01\) | \(0.2968\) | \(0.744\) | \(1.04\) | \(1.40\) | \(133\) | \(35.0\) |
| Ethane | \(\mathrm{C}_2 \mathrm{H}_6\) | \(30.07\) | \(0.2765\) | \(1.48\) | \(1.75\) | \(1.18\) | \(305.4\) | \(48.8\) |
| Ethylene | \(\mathrm{C}_2 \mathrm{H}_4\) | \(28.05\) | \(0.2964\) | \(1.23\) | \(1.53\) | \(1.24\) | \(283\) | \(51.2\) |
| Helium | \(\mathrm{He}\) | \(4.003\) | \(2.077\) | \(3.12\) | \(5.19\) | \(1.67\) | \(5.2\) | \(2.3\) |
| Hydrogen | \(\mathrm{H}_2\) | \(2.016\) | \(4.124\) | \(10.2\) | \(14.3\) | \(1.40\) | \(33.2\) | \(13.0\) |
| Methane | \(\mathrm{CH}_4\) | \(16.04\) | \(0.5183\) | \(1.70\) | \(2.22\) | \(1.31\) | \(190.7\) | \(46.4\) |
| Nitrogen | \(\mathrm{N}_2\) | \(28.01\) | \(0.2968\) | \(0.743\) | \(1.04\) | \(1.40\) | \(126.2\) | \(33.9\) |
| Oxygen | \(\mathrm{O}_2\) | \(32.00\) | \(0.2598\) | \(0.658\) | \(0.918\) | \(1.40\) | \(154.4\) | \(50.5\) |
| Propane | \(\mathrm{C}_3 \mathrm{H}_8\) | \(44.09\) | \(0.1886\) | \(1.48\) | \(1.67\) | \(1.13\) | \(370\) | \(42.5\) |
| Refrigerant 134a | \(\mathrm{C}_2 \mathrm{F}_4 \mathrm{H}_2\) | \(102.03\) | \(0.08149\) | \(0.76\) | \(0.85\) | \(1.12\) | \(374.3\) | \(40.6\) |
| Water (Steam) | \(\mathrm{H}_2 \mathrm{O}\) | \(18.02\) | \(0.4614\) | \(1.40\) | \(1.86\) | \(1.33\) | \(647.3\) | \(220.9\) |
| Liquids | Solids * | |||||
|---|---|---|---|---|---|---|
| Substance | Temp \(({ }^{\circ} \mathrm{C})\) | \( \dfrac{\rho}{\left[ \dfrac{\mathrm{kg}}{\mathrm{m}^3}\right]} \) | \(\dfrac{c_{P}}{\left[ \dfrac{\mathrm{kJ}}{\mathrm{kg} \cdot \mathrm{K}}\right]}\) | Substance | \( \dfrac{\rho}{\left[ \dfrac{\mathrm{kg}}{\mathrm{m}^3}\right]} \) | \(\dfrac{c_{P}}{\left[ \dfrac{\mathrm{kJ}}{\mathrm{kg} \cdot \mathrm{K}}\right]}\) |
| Ammonia | \(25\) | \(602\) | \(4.80\) | Aluminum | \(2,700\) | \(0.902\) |
| Benzene | \(20\) | \(879\) | \(1.72\) | Brass, yellow | \(8,310\) | \(0.400\) |
| Brine \(( 20 / % \mathrm{NaCl})\) | \(20\) | \(1,150\) | \(3.11\) | Brick (common) | \(1,922\) | \(0.79\) |
| Ethanol | \(25\) | \(783\) | \(2.46\) | Concrete | \(2,300\) | \(0.653\) |
| Ethyl alcohol | \(20\) | \(789\) | \(2.84\) | Copper | \(8,900\) | \(0.386\) |
| Ethylene glycol | \(20\) | \(1,109\) | \(2.84\) | Glass, window | \(2,700\) | \(0.800\) |
| Kerosene | \(20\) | \(820\) | \(2.00\) | Iron | \(7,840\) | \(0.45\) |
| Mercury | \(25\) | \(13,560\) | \(0.139\) | Lead | \(11,310\) | \(0.128\) |
| Oil (light) | \(25\) | \(910\) | \(1.80\) | Silver | \(10,470\) | \(0.235\) |
| Refrigerant 134a | \(25\) | \(1,206\) | \(1.42\) | Steel (mild) | \(7,830\) | \(0.500\) |
| Water | \(25\) | \(997\) | \(4.18\) | * Evaluated at room temperature. | ||
| Values adapted from K. Wark, Jr. and D. E. Richards, Thermodynamics, 6th ed. (McGraw-Hill, New York, 1999) and Y. A. Cengul and M. A. Boles, Thermodynamics, 4th ed. (McGraw-Hill, New York, 2002). | ||||||
| Gases (at \(77 ^{\circ} \mathrm{F}\) and \(1 \mathrm{~atm}\)) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Substance | Molar Mass | \(\dfrac{R}{\left[ \dfrac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{lbm} \cdot { }^{\circ} \mathrm{R}}\right]}\) | \(\dfrac{c_{v}}{\left[ \dfrac{\mathrm{Btu}}{\mathrm{lbm} \cdot { }^{\circ} \mathrm{R}}\right]}\) | \(\dfrac{c_{P}}{\left[ \dfrac{\mathrm{Btu}}{\mathrm{lbm} \cdot { }^{\circ} \mathrm{R}}\right]}\) | \(k\) | \(\dfrac{T_{c}}{ \text{}^{\circ} \mathrm{R}}\) | \(\dfrac{P_{c}}{\mathrm{atm}}\) | |
| Acetylene | \(\mathrm{C}_2 \mathrm{H}_2\) | \(26.04\) | \(59.33\) | \(0.328\) | \(0.404\) | \(1.23\) | \(556\) | \(61.6\) |
| Air | --- | \(28.97\) | \(59.33\) | \(0.171\) | \(0.240\) | \(1.40\) | \(239\) | \(37.2\) |
| Ammonia | \(\mathrm{NH}_3\) | \(17.04\) | \(90.67\) | \(0.397\) | \(0.514\) | \(1.30\) | \(730\) | \(111.3\) |
| Carbon dioxide | \(\mathrm{CO}_2\) | \(44.01\) | \(35.11\) | \(0.156\) | \(0.202\) | \(1.29\) | \(548\) | \(72.9\) |
| Carbon monoxide | \(\mathrm{CO}\) | \(28.01\) | \(55.16\) | \(0.178\) | \(0.249\) | \(1.40\) | \(239\) | \(34.5\) |
| Ethane | \(\mathrm{C}_2 \mathrm{H}_6\) | \(30.07\) | \(51.38\) | \(0.353\) | \(0.419\) | \(1.19\) | \(549\) | \(48.2\) |
| Ethylene | \(\mathrm{C}_2 \mathrm{H}_4\) | \(28.05\) | \(55.08\) | \(0.294\) | \(0.365\) | \(1.24\) | \(510\) | \(50.5\) |
| Helium | \(\mathrm{He}\) | \(4.003\) | \(386.0\) | \(0.744\) | \(1.24\) | \(1.67\) | \(9.3\) | \(2.26\) |
| Hydrogen | \(\mathrm{H}_2\) | \(2.016\) | \(766.4\) | \(2.43\) | \(3.42\) | \(1.40\) | \(59.8\) | \(12.8\) |
| Methane | \(\mathrm{CH}_4\) | \(16.04\) | \(96.32\) | \(0.407\) | \(0.531\) | \(1.30\) | \(344\) | \(45.8\) |
| Nitrogen | \(\mathrm{N}_2\) | \(28.01\) | \(55.16\) | \(0.178\) | \(0.248\) | \(1.39\) | \(227\) | \(33.5\) |
| Oxygen | \(\mathrm{O}_2\) | \(32.00\) | \(48.28\) | \(0.157\) | \(0.219\) | \(1.40\) | \(278\) | \(49.8\) |
| Propane | \(\mathrm{C}_3 \mathrm{H}_8\) | \(44.09\) | \(35.04\) | \(0.355\) | \(0.400\) | \(1.13\) | \(666\) | \(42.1\) |
| Refrigerant 134a | \(\mathrm{C}_2 \mathrm{F}_4 \mathrm{H}_2\) | \(102.03\) | \(15.14\) | \(0.184\) | \(0.203\) | \(1.10\) | \(672.8\) | \(40.1\) |
| Water (Steam) | \(\mathrm{H}_2 \mathrm{O}\) | \(18.02\) | \(87.74\) | \(0.335\) | \(0.445\) | \(1.33\) | \(1165\) | \(218.0\) |
| Liquids | Solids * | |||||
|---|---|---|---|---|---|---|
| Substance | Temp \(({ }^{\circ} \mathrm{F})\) | \( \dfrac{\rho}{\left[ \dfrac{\mathrm{lbm}}{\mathrm{ft}^3}\right]} \) | \(\dfrac{c_{P}}{\left[ \dfrac{\mathrm{Btu}}{\mathrm{lbm} \cdot { }^{\circ} \mathrm{R}}\right]}\) | Substance | \( \dfrac{\rho}{\left[ \dfrac{\mathrm{lbm}}{\mathrm{ft}^3}\right]} \) | \(\dfrac{c_{P}}{\left[ \dfrac{\mathrm{Btu}}{\mathrm{lbm} \cdot { }^{\circ} \mathrm{R}}\right]}\) |
| Ammonia | \(80\) | \(37.5\) | \(1.135\) | Aluminum | \(170\) | \(0.215\) |
| Benzene | \(68\) | \(54.9\) | \(0.411\) | Brass, yellow | \(519\) | \(0.0955\) |
| Brine \(( 20 / % \mathrm{NaCl})\) | \(68\) | \(71.8\) | \(0.743\) | Brick (common) | \(120\) | \(0.189\) |
| Ethanol | \(77\) | \(48.9\) | \(0.588\) | Concrete | \(144\) | \(0.156\) |
| Ethyl alcohol | \(68\) | \(49.3\) | \(0.678\) | Copper | \(555\) | \(0.0917\) |
| Ethylene glycol | \(68\) | \(69.2\) | \(0.678\) | Glass, window | \(169\) | \(0.191\) |
| Kerosene | \(68\) | \(51.2\) | \(0.478\) | Iron | \(490) | \(0.107\) |
| Mercury | \(77\) | \(847\) | \(0.033\) | Lead | \(705\) | \(0.030\) |
| Oil (light) | \(77\) | \(56.8\) | \(0.430\) | Silver | \(655\) | \(0.056\) |
| Refrigerant 134a | \(32\) | \(80.9\) | \(0.318\) | Steel (mild) | \(489\) | \(0.119\) |
| Water | \(68\) | \(62.2\) | \(1.00\) | * Evaluated at room temperature. | ||
| Values adapted from K. Wark, Jr. and D. E. Richards, Thermodynamics, 6th ed. (McGraw-Hill, New York, 1999) and Y. A. Cengul and M. A. Boles, Thermodynamics, 4th ed. (McGraw-Hill, New York, 2002). | ||||||
A rigid tank contains \(0.80 \mathrm{~g}\) of air initially at \(295 \mathrm{~K}\) and \(1.5 \mathrm{~bars}\) and an electric resistor wire. The electric resistor within the tank has a mass of \(0.05 \mathrm{~g}\) and is energized by passing a current of \(0.6 \mathrm{~A}\) for \(30 \mathrm{~s}\) from a \(12.0 \mathrm{V}\) source. During the same time interval, \(156 \mathrm{~J}\) of energy is lost through the walls of the tank by heat transfer. Assume that the air can be modeled as an ideal gas with room temperature specific heats and the mild steel resistance wire can be modeled as an incompressible substance with room temperature specific heats.
Determine (a) the final temperature of the gas, in kelvins, and (b) the final pressure of the gas, in bars.
Solution
Known: An electric resistor is used to heat air contained in a rigid tank.
Find: (a) The final temperature of the gas, in \(\mathrm{K}\).
(b) The final pressure of the gas, in bars.
Given:
.png?revision=1)
Figure \(\PageIndex{1}\): An electrical wire passes into a rigid container of air, containing a resistor.
Analysis:
Strategy \(\rightarrow\) Because we are dealing with heat transfer and electrical work and we are asked to find the final temperature, use conservation of energy. (Questions involving changes in pressure and temperature of a substance frequently require the use of conservation of energy and some equation of state to describe how the thermophysical properties of the substance are related.)
System \(\rightarrow\) Because they told us about the heat transfer from the gas to the tank, let's treat everything inside the tank, gas and resistor, as a closed system. (See dashed line drawn in figure above.)
Property to count \(\rightarrow\) We want to know temperature but energy is the property we have a conservation principle for and we know energy and temperature are related, so lets count energy.
Time interval \(\rightarrow\) Finite-time form since they give us the 30-second time interval.
Because we may not recall the finite-time form for a closed system, let's quickly redevelop it: \[\begin{aligned} &\frac{d E_{\text{sys}}}{dt} = \dot{Q}_{\text{net, in}} + \dot{W}_{\text{net, in}} + \underbrace{ \cancel{\sum_{\text{in}} \dot{m}_{i} \left(h_{i}+\frac{V_{i}^{2}}{2}+g z\right) - \sum_{\text{out}} \dot{m}_{e} \left(h_{e}+\frac{V_{e}^{2}}{2}+g z\right)}^{=0} }_{\text {Closed system}} \\ & \int\limits_{t_{1}}^{t_{2}} \left(\frac{d E_{\text{sys}}}{dt}\right) dt = \int\limits_{t_{1}}^{t_{2}} \left(\dot{Q}_{\text {net, in}} + \dot{W}_{\text {net, in}}\right) dt \quad \rightarrow \quad \boxed{\Delta E_{\text{sys}} = Q_{\text {net, in}} + W_{\text {net, in}}} \end{aligned} \nonumber \]
Now that we have the correct form of the energy balance, we need to evaluate the various terms as below: \[\underbrace{ \cancel{\Delta E_{\text{sys}}}^{= \Delta U_{\text{sys}}} }_{\begin{array}{c} \text{No changes in kinetic} \\ \text{and gravitational \\ \text{potential energy} \end{array}} = \underbrace{ \cancel{Q_{\text {net, in}}}^{=-Q_{\text {out}}} }_{\begin{array}{c} \text{Use given information} \\ \text{about heat transfer out} \\ \text{of the system} \end{array}} +\underbrace{ \cancel{W_{\text {net, in}}}^{=W_{\text {electric, in}}} }_{\begin{array}{c} \text{Only one type of work} \\ \text{because the system} \\ \text{is contained in a rigid tank.} \end{array}} \quad\quad \rightarrow \quad\quad \Delta U_{\text {sys}} = -Q_{\text {out}} + W_{\text {electric, in}} \nonumber \]
First, let's examine the change in internal energy of the system. We must recognize that the change in internal energy for the whole system can be calculated as the sum of the change in internal energy for each of its subsystems. (Recall this is a key feature of an extensive property like energy.) \[\Delta U_{\text{sys}} = \Delta U_{\text{gas}} + \Delta U_{\text{resistor}} = m_{\text{gas}} \Delta u_{\text{gas}} + m_{\text{resistor}} \Delta u_{\text{resistor}} \nonumber \]
\[ \begin{aligned} &\text{but } \quad &\Delta u_{\text{gas}} = c_{v, \text { gas}} \left(T_{2}-T_{1}\right)_{\text {gas}} \quad\quad &| \text{ Ideal gas with room temperature specific heats} \\ &\text{and } \quad &\Delta u_{\text{resistor}} = c_{\text{resistor}} \left(T_{2}-T_{1}\right)_{\text{resistor}} \quad &| \text{ Incomp. substance with room temperature specific heats} \end{aligned} \nonumber \]
\[ \text{So,} \quad\quad \Delta U_{\text{sys}} = \left[m c_{v} \left(T_{2}-T_{1}\right)\right]_{\text{gas}} + \left[m c \left(T_{2}-T_{1}\right)\right]_{\text{resistor}} \nonumber \]
Second, we must evaluate the electric work. We can recover the necessary equation from the definition of electrical power: \[\begin{aligned} W_{\text {electric, in}} &= \int\limits_{t_{1}}^{t_{2}} \dot{W}_{\text {electric, in}} \ dt = \int\limits_{t_{1}}^{t_{2}}(i \Delta V) dt = (i \Delta V) \int\limits_{t_{1}}^{t_{2}} dt = (i \Delta V) \Delta t \\ &= (0.60 \mathrm{~A}) \cdot (12.0 \mathrm{~V}) \cdot (30 \mathrm{~s}) = 216 \mathrm{~W} \cdot \mathrm{s} = 216 \mathrm{~J} \end{aligned} \nonumber \] Combining all of this information we have the following result: \[\Delta U_{\text{sys}} = -Q_{\text {out}} + W_{\text {electric, in}} \quad \rightarrow \quad \left[m c_{v}\left(T_{2}-T_{1}\right)\right]_{\text{gas}} + \left[m c \left(T_{2}-T_{1}\right)\right]_{\text{resistor}} = [-156+216] \mathrm{~J} \nonumber \]
Before we can solve for the temperatures, we must make an assumption about the temperatures of the gas and the resistor. It would seem reasonable that the resistor and the gas have the same temperatures if the system is in thermal equilibrium at beginning and end. In addition, we must also find room temperature values for the specific heats by consulting the appropriate tables: \(c_{v}, \text{ gas} = 0.718 \mathrm{~kJ} /(\mathrm{kg} \cdot\mathrm{K})\) and \(c_{\text {resistor}} = 0.500 \mathrm{~kJ} /(\mathrm{kg} \cdot \mathrm{K})\).
Using this in the energy balance we can now solve for the final temperature as follows: \[\begin{gathered} {\left[mc_{v} \left(T_{2}-T_{1}\right)\right]_{\text{gas}} + \left[m c\left(T_{2}-T_{1}\right)\right]_{\text{resistor}} = [-156+216] \mathrm{~J}} \\[4pt] \left(m_{\text {gas }} c_{v, \text { gas}} + m_{\text {resistor }} c_{\text {resistor}}\right)\left(T_{2}-T_{1}\right) = 60 \mathrm{~J} \quad \rightarrow \quad \mathrm{T}_{2}-T_{1} = \frac{60 \mathrm{~J}}{\left(m_{\text {gas }} c_{v, \text { gas}} + m_{\text {resistor }} c_{\text {resistor}}\right)} \\[4pt] T_{2}-T_{1}=\frac{60 \mathrm{~J}}{\left[(0.80 \mathrm{~g}) \cdot \left(0.718 \ \dfrac{\mathrm{J}}{\mathrm{g} \cdot \mathrm{K}}\right) + (0.05 \mathrm{~g}) \cdot \left(0.500 \ \dfrac{\mathrm{J}}{\mathrm{g} \cdot \mathrm{K}}\right)\right]} = \frac{60 \mathrm{~J}}{\left[(0.574+0.025) \ \dfrac{\mathrm{J}}{\mathrm{K}}\right]} = 100 \mathrm{~K} \\[4pt] T_{2}=T_{1}+100 \mathrm{~K} = 295 \mathrm{~K} + 100 \mathrm{~K} = 395 \mathrm{~K} \end{gathered} \nonumber \]
Solving for the final pressure is done by applying the ideal gas equation as follows: \[\begin{gathered} \left.\begin{array}{l} P_{1} V\kern-1.0em\raise0.3ex-_{1} = m_{1} R_{\text {air }} T_{1} \\ P_{2} V\kern-1.0em\raise0.3ex-_{2} = m_{2} R_{\text {air }} T_{2} \end{array}\right\} \quad\rightarrow\quad \frac{P_{1} V\kern-0.8em\raise0.3ex-_{1}}{P_{2} V\kern-0.8em\raise0.3ex-_{2}} = \frac{m_{1} R_{\text {air }} T_{1}}{m_{2} R_{\text {air }} T_{2}} \quad\rightarrow\quad \left(\frac{P_{1}}{P_{2}}\right) \cancel{\left(\frac{V\kern-0.8em\raise0.3ex-_{1}}{V\kern-0.8em\raise0.3ex-_{2}}\right)}^{=1} = \cancel{\left(\frac{m_{1}}{m_{2}}\right)}^{=1} \cdot \cancel{\left(\frac{R_{\text{air}}}{R_{\text {air}}}\right)}^{=1} \cdot \left(\frac{T_{1}}{T_{2}}\right) \\{4pt} P_{2} = P_{1}\left(\frac{T_{2}}{T_{1}}\right) = (1.5 \text { bars})\left(\frac{395 \mathrm{~K}}{295 \mathrm{~K}}\right) = 2.01 \text { bars} \end{gathered} \nonumber \]
Comment:
(1) In applying the energy balance, we went from \(\Delta U\) to \(m \Delta u\). This is necessary because our substance models only allow us to calculate the change in specific internal energy: \(\Delta u\), not \(\Delta U\). Furthermore, this is how temperature enters the picture.
(2) When applying the ideal gas equation to find the pressure notice how we made use of ratios. This greatly simplifies the calculations and allows us to easily handle different units. Instead of solving symbolically and recognizing the ratios the final pressure could have been obtained as follows:
Step 1: Solve for \(V\kern-1.0em\raise0.3ex-_{1} \quad V\kern-1.0em\raise0.3ex-_{1} = \dfrac{m_{1} R_{\text {air }} T_{1}}{P_{1}}\)
Step 2: Recognize that \(V\kern-1.0em\raise0.3ex-_{2} = V\kern-0.8em\raise0.3ex-_{1}\) and that \(m_{2}=m_{1}\)
Step 3: Solve for \(P_{2} \quad P_{2}=\dfrac{m_{2} R_{\text {air }} T_{2}}{V_{2}} = \dfrac{m_{1} R_{\text {air }} T_{2}}{V_{1}}\)
Ratios can greatly speed-up and simplify calculations. In addition, it reduces errors, e.g. avoiding the need to find \(R_{\mathrm{gas}}\) in units that work with bars plus the extra number punching on your calculator.
(3) If we had not made the assumption that the resistor and the gas had equal temperatures it would have been impossible to solve the problem without additional assumptions or information.
(4) What heat transfer and work interactions occurred between the resistor and its surroundings during this process? Between the gas and its surroundings?
A pump is used to move kerosene between two points in a piping system. The pump is located between the two points. Kerosene enters the piping system at an elevation of \(5 \mathrm{~ft}\), a pressure of \(15 \mathrm{~psia}\) and a temperature of \(70^{\circ} \mathrm{F}\) and leaves the piping system at an elevation of \(20 \mathrm{~ft}\) and a pressure of \(60 \mathrm{~psia}\). During the adiabatic pumping process, the kerosene experiences a \(0.5^{\circ} \mathrm{F}\) rise in temperature. Determine the power required to operate the pump in \(\mathrm{ft} \cdot \mathrm{lbf} / \mathrm{lbm}\).
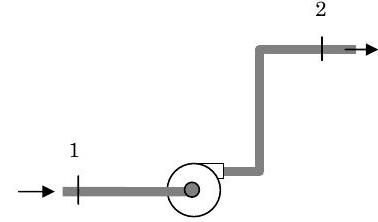
Figure \(\PageIndex{2}\): Kerosene is pumped to a higher elevation.
Solution
Known: Kerosene is pumped steadily in a piping system
Find: The power required to operate the pump in \(\mathrm{ft} \cdot \mathrm{lbf} / \mathrm{lbm}\).
Given:
Kerosene State 1: \(z_{1}=5 \mathrm{ft}; \ P_{1}=15 \mathrm{~psia} ; \ T_{1}=70^{\circ} \mathrm{F}\)
Kerosene State 2: \(z_{2}=20 \mathrm{ft}; \ P_{2}=60 \mathrm{~psia}\)
Process 1-2: Adiabatic, steady-state
\(T_{2}-T_{1}=0.5^{\circ} \mathrm{F}\)
Analysis:
Strategy \(\rightarrow\) Use conservation of energy
System \(\rightarrow\) Non-deforming open system that includes pipe, pump, and contents.
Property \(\rightarrow\) Energy (and possibly mass)
Time interval \(\rightarrow\) Since steady-state, infinitesimal time period.
Writing the energy balance for the open system gives the following: \[ \begin{aligned} \underbrace{ \cancel{\frac{d E_{\text{sys}}}{dt}}^{=0} }_{\begin{array}{c} \text{Steady-state} \\ \text{conditions} \end{array}} = \underbrace{ \cancel{\dot{Q}_{\text{net, in}}}^{=0} }_{\text{Adiabatic}} + \dot{W}_{\text{net, in}} + \dot{m}_{1} \left(h_{1} \frac{V_{1} { }^{2}}{2} + gz_{1}\right) - \dot{m}_{2} \left(h_{2} \frac{V_{2} { }^{2}}{2} + gz_{2}\right) \\ 0 &= \dot{W}_{\text{pump, in}} \dot{m}_{1} \left(h_{1} \frac{V_{1} { }^{2}}{2} + gz_{1}\right) - \dot{m}_{2} \left(h_{2} \frac{V_{2} { }^{2}}{2} + gz_{2}\right) \end{aligned} \nonumber \]
To go further we must say something about the mass flow rates. If we apply the conservation of mass to this steady-state, one-inlet/one-outlet system, we find that the mass flow rates are equal. Using this result, the equation above becomes: \[\frac{\dot{W}_{\text {pump, in}}}{\dot{m}} = \left(h_{2}-h_{1}\right) + \left(\frac{V_{2}{ }^{2}}{2}-\frac{V_{1}{ }^{2}}{2}\right) + g\left(z_{2}-z_{1}\right) \nonumber \] The term on the left-hand side is the quantity we are looking for. It is the power per unit mass flow rate or the work per unit mass. (As stated earlier, it is very common to solve open system problems on a per-unit-mass basis. Then if we only change the mass flow rate we do not have to resolve the problem, unless something else changes.)
The change in specific enthalpy can be handled by assuming that kerosene can be modeled as an incompressible substance with room temperature specific heats; thus, \[h_{2}-h_{1} = u_{2}-u_{1} + \upsilon\left(P_{2}-P_{1}\right) = c\left(T_{2}-T_{1}\right) + \upsilon\left(P_{2}-P_{1}\right) \nonumber \] where \(c=0.478 \mathrm{~Btu} / \left(\mathrm{lbm}{ }^{\circ} \mathrm{R}\right)\) and \(\upsilon = 1 / \rho=1 /\left(51.2 \mathrm{~lbm} / \mathrm{ft}^{3}\right)\)
We have no information about the velocity of the kerosene at either the inlet or outlet. Without making some assumption about the velocities we cannot solve for the power. We need not assume absolute values for the velocity but only that its change—actually, the change in kinetic energy—is negligible.
Using these results we can now solve for the power: \[\begin{aligned} \frac{\dot{W}_{\text {pump, in}}}{\dot{m}} &= \left(h_{2}-h_{1}\right) + \underbrace{ \cancel{\left{\frac{V_{2}{ }^{2}}{2} - \frac{V_{1}{ }^{2}}{2}\right)}^{=0} }_{\begin{array}{c} \text{Assume change is} \\ \text{negligible} \end{array}} + g\left(z_{2}-z_{1}\right) \\ &=c\left(T_{2}-T_{1}\right)+v\left(P_{2}-P_{1}\right)+g\left(z_{2}-z_{1}\right)=c\left(T_{2}-T_{1}\right)+\frac{\left(P_{2}-P_{1}\right)}{\rho}+g\left(z_{2}-z_{1}\right) \end{aligned} \nonumber \]
Now we solve for the individual terms in the energy balance as follows: \[c\left(T_{2}-T_{1}\right) = \left(0.478 \ \frac{\mathrm{Btu}}{\mathrm{lbm} \cdot{ }^{\circ} \mathrm{R}}\right) \left(0.5^{\circ} \mathrm{F}\right) = 0.239 \ \frac{\mathrm{Btu}}{\mathrm{lbm}} = \left(0.239 \ \frac{\mathrm{Btu}}{\mathrm{lbm}}\right) \times \left(778 \ \frac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{Btu}}\right) = 185.9 \ \frac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{lbm}} \nonumber \]
where we recognize the \({ }^{\circ} \mathrm{R}\) and the \({ }^{\circ} \mathrm{F}\) cancel, because they both represent temperature differences and not temperatures: \[\begin{aligned} &\frac{\left(P_{2}-P_{1}\right)}{\rho} = \frac{(60-15) \mathrm{~psia}}{51.2 \ \dfrac{\mathrm{lbm}}{\mathrm{ft}^{3}}} = 0.8789 \ \frac{\mathrm{psia} \cdot \mathrm{ft}^{3}}{\mathrm{lbm}} = \left(0.8789 \ \frac{\mathrm{psia} \cdot \mathrm{ft}^{3}}{\mathrm{lbm}}\right) \times \left(\frac{\mathrm{lbf} / \mathrm{in}^{2}}{\mathrm{psia}}\right) \times \left(\frac{144 \mathrm{~in}^{2}}{1 \mathrm{~ft}^{2}}\right) = 126.6 \ \frac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{lbm}} \\ { } \\ & g\left(z_{2}-z_{1}\right) = \left(32.174 \ \frac{\mathrm{ft}}{\mathrm{s}^{2}}\right) \cdot [(20-5) \mathrm{~ft}] = 482.6 \ \frac{\mathrm{ft}^{2}}{\mathrm{~s}^{2}} = \left(482.6 \ \frac{\mathrm{ft}^{2}}{\mathrm{~s}^{2}}\right) \times \left(\frac{\mathrm{lbm}}{\mathrm{lbm}}\right) \times \left(\frac{1 \mathrm{~lbf}}{32.174 \ \dfrac{\mathrm{lbm} \cdot \mathrm{ft}}{\mathrm{s}^{2}}}\right) = 15.0 \ \frac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{lbm}} \end{aligned} \nonumber \]
Combining these results give \[\frac{\dot{W}_{\text {pump, in}}}{\dot{m}} = \underbrace{c\left(T_{2}-T_{1}\right)}_{=57 \%} + \underbrace{\frac{1}{\rho}\left(P_{2}-P_{1}\right)}_{=39 \%} + \underbrace{g\left(z_{2}-z_{1}\right)}_{=4 \%} = (185.9+126.6+15.0) \ \frac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{lbm}} = 328 \ \frac{\mathrm{ft} \cdot \mathrm{lbf}}{\mathrm{lbm}} \nonumber \]
Comments:
(1) Note how the power breaks down. Assuming that increasing the pressure and elevation are the desired effects for a pump, approximately \(57 \%\) of the energy is "wasted" in the \(0.5^{\circ} \mathrm{F}\) temperature change in the liquid. What would the power be if the process had been isothermal?
(2) What if the only change in the problem had been that the temperature of the kerosene decreased by \(0.5^{\circ} \mathrm{F}\)? What would the power input have been?
[This is pretty impressive. First, you hook a pipe up to some kerosene at room conditions. Then it increases the elevation and pressure of the kerosene, "cools" the kerosene in an adiabatic process, and gives you some power out. You could probably get the high-pressure, elevated kerosene to flow through a hydraulic turbine and get even more work out. As appealing as this might be, we will soon discover that it would be impossible! If you figure out a way to do this, please don't tell anyone but contact your instructor and you can both retire to the Bahamas!]
(3) Now, if the pump had really been a compressor and instead of kerosene we were compressing air, what would the power have been? (Assume changes in kinetic energy are negligible.) Does the pressure even fit into your calculations? [Answer: \(108 \mathrm{~ft} \cdot \mathrm{lbf} / \mathrm{lbm}\) ]
[We will shortly discover that this process is also impossible. Under the most ideal conditions, the air temperature would increase approximately \(258^{\circ} \mathrm{F}\) if it was compressed adiabatically between the given inlet state and final pressure.]
Tank A contains \(1.0 \mathrm{~kg}\) of nitrogen \(\left(\mathrm{N}_{2}\right)\) gas initally at \(150 \mathrm{~kPa}\) and \(300 \mathrm{~K}\). Attached to this tank through a suitable valve is a second tank B that contains \(2.0 \mathrm{~kg}\) of the same gas at \(300 \mathrm{~kPa}\) and \(400 \mathrm{~K}\). Both tanks are rigid and insulated. Assume that nitrogen gas can be modeled as an ideal gas with room temperature specific heats.
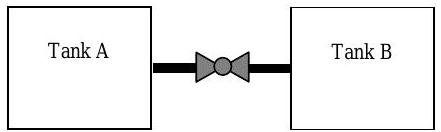
Figure \(\PageIndex{3}\): Setup of two connected tanks.
If the valve is opened and equilibrium is reached, determine
(a) the final mixture temperature, in \(\mathrm{K}\), and
(b) the final pressure of the mixture, in \(\mathrm{kPa}\).
Solution
Known: The contents of two tanks each containing nitrogen are mixed together.
Find: (a) final temperature of the mixture, in \(\mathrm{K}\), and (b) final pressure of the mixture in \(\mathrm{kPa}\).
Given:
| Initial State (1) | Final State (2) | |||
|---|---|---|---|---|
| Tank A | Tank B | Tank A | Tank B | |
| \[\begin{gathered} 1.0 \mathrm{~kg} \\ 150 \mathrm{~kPa} \\ 300 \mathrm{~K} \end{gathered} \nonumber \] | \[\begin{gathered} 2.0 \mathrm{~kg} \\ 300 \mathrm{~kPa} \\ 400 \mathrm{~K} \end{gathered} \nonumber \] | \(T_{2} \text{ and } P_{2}\) | \(T_{2} \text{ and } P_{2}\) | |
Analysis:
Strategy \(\rightarrow\) Conservation of energy
System \(\rightarrow\) Treat the contents of both tanks as a single closed system.
Property to count \(\rightarrow\) Energy and mass
Time interval \(\rightarrow\) Finite-time since beginning information given and ending information desired.
Writing the finite-time form of the conservation of energy for a closed system we have the following: \[ \underbrace{ \cancel{\Delta E_{\text{sys}}}^{=\Delta U_{\text{sys}}} }_{\begin{array}{c} \text{Changes in } E_{K} \text{ and} \\ E_{GP} \text{ are negligible} \end{array}} = \underbrace{ \cancel{Q_{\text{net, in}}}^{=0} }_{\begin{array}{c} \text{Tanks specified} \\ \text{as adiabatic} \end{array}} + \underbrace{ \cancel{W_{\text{net, in}}}^{=0} }_{\begin{array}{c} \text{Rigid tanks and no other} \\ \text{work interactions identified} \end{array}} \quad \rightarrow \quad \Delta U_{\text{sys}} = U_{\text{sys, } 2} - U_{\text{sys, } 1} = 0 \nonumber \]
Now to evaluate the internal energies of the closed system gives the following, assuming the air can be modeled as an ideal gas with room temperature specific heats:
\[\begin{aligned} 0 &= U_{\text{sys,} 2}-U_{\text{sys,} 1} \\[4pt] &=\left(m_{\text{sys}} u\right)_{2} - \left(m_{A} u_{A}+m_{B} u_{B}\right)_{1} \quad \text { but } \quad m_{\text{sys}} = m_{A, 2}+m_{B, 2}=m_{A, 1}+m_{B, 1} \\[4pt] &= \left[\left(m_{A, 1}+m_{B, 1}\right) u_{2}\right]-\left[m_{A, 1} u_{A, 1}+m_{B, 1} u_{B, 1}\right] \\[4pt] &=m_{A, 1}\left(u_{2}-u_{A, 1}\right)+m_{B, 1}\left(u_{2}-u_{B, 1}\right) \quad\quad\quad \mid \text { collecting the } m_{A} \text { and } m_{B} \text { terms to get } \Delta u \\[4pt] &=m_{A, 1} c_{v}\left(T_{2}-T_{A, 1}\right)+m_{B, 1} c_{v}\left(T_{2}-T_{B, 1}\right) \quad\quad \mid \text { applying the ideal gas model } \end{aligned} \nonumber \]
Because our ideal gas model assumes that the specific heats are all evaluated at the room temperature, they are constant and will cancel out when we solve for the final temperature: \[\begin{gathered} 0=m_{A, 1} c_{v} \left(T_{2}-T_{A, 1}\right) + m_{B, 1} c_{v}\left(T_{2}-T_{B, 1}\right) \quad \rightarrow \quad T_{2} = \frac{m_{A, 1} c_{v} T_{A, 1}+m_{B, 1} c_{v} T_{B, 1}}{m_{A, 1} c_{v}+m_{B, 1} c_{v}} = \frac{m_{A, 1} T_{A, 1}+m_{B, 1} T_{B, 1}}{m_{A, 1}+m_{B, 1}} \\[4pt] T_{2}=\frac{(1.0 \mathrm{~kg})(300 \mathrm{~K})+(2.0 \mathrm{~kg})(400 \mathrm{~K})}{(1.0+2.0) \mathrm{~kg}} = 367 \mathrm{~K} \end{gathered} \nonumber \]
Now to solve for the final pressure, we apply the ideal gas model: \[\begin{aligned} \left.\begin{array}{l} P_{2}=\dfrac{m_{2} R_{\text {air }} T_{2}}{V\kern-1.0em\raise0.3ex-_{2}} \\[4pt] V\kern-1.0em\raise0.3ex-_{2} = V\kern-1.0em\raise0.3ex-_{A}+V\kern-0.8em\raise0.3ex-_{B} \\[4pt] V\kern-1.0em\raise0.3ex-_{A}=\dfrac{m_{A, 1} R_{\text {air }} T_{A, 1}}{P_{A, 1}} \\[4pt] V\kern-1.0em\raise0.3ex-_{B}=\dfrac{m_{B, 1} R_{\text {air }} T_{B, 1}}{P_{B, 1}} \end{array}\right\} \quad \rightarrow \quad & P_{2}=\frac{m_{2} R_{\text {air }} T_{2}}{V\kern-0.8em\raise0.3ex-_{2}} = \frac{m_{1} R_{\text {air }} T_{2}}{V\kern-1.0em\raise0.3ex-_{A}+V\kern-0.8em\raise0.3ex-_{B}} = \frac{\left(m_{A, 1}+m_{B, 1}\right) R_{\text {air }} T_{2}}{\dfrac{m_{A, 1} R_{\text {air }} T_{A, 1}}{P_{A, 1}}+\dfrac{m_{B, 1} R_{\text {air }} T_{B, 1}}{P_{B, 1}}} = \frac{\left(m_{A, 1}+m_{B, 1}\right) T_{2}}{\dfrac{m_{A, 1} T_{A, 1}}{P_{A, 1}}+\dfrac{m_{B, 1} T_{B, 1}}{P_{B, 1}}} \\ & P_{2}=\frac{(1.0+2.0)(367)}{(1.0)\left(\dfrac{300}{150 \mathrm{~kPa}}\right) + (2.0)\left(\dfrac{400}{300 \mathrm{~kPa}}\right)} = 236 \mathrm{~kPa} \end{aligned} \nonumber \]
Comment:
(1) Notice how we once again have used the idea of ratios to solve for the final pressure without ever calculating the tank volumes. Obviously, as a check you could find the volume of each tank, add them to find the total volume, and then use the ideal gas equation. This would be a good check.
(2) Under our model for ideal gases, the specific heat value cancels out. With a more accurate model to handle larger temperature changes, the specific heat values would not drop out of the process. Note that the final temperature is not just the average of the initial temperatures.
The substance models introduced in this section have only limited validity. Under certain conditions, the kerosene that we pumped might in fact start to vaporize or boil. Similarly under certain conditions when a gas is compressed, liquid droplets might condense or solid particles might precipitate out. Changes in phase occur suddenly and nothing about our model allows us to predict when this will occur. To learn more about these important phenomena, we will study the behavior of real substances in a later course (ES202 — Fluid & Thermal Systems). Once we have done this we will have a better basis for deciding when our substance models are accurate.
As an indication of these limitations, the ideal gas model is typically assumed to be valid at "high" temperatures and "low" pressures. But how high is "high" and how low is "low"? Well, the last two columns of the thermophysical property data for gases give us some guidance. The points of reference for gases are the critical pressure \(P_{\mathrm{c}}\) and the critical temperature \(T_{\mathrm{c}}\). As we will learn later, the ideal gas equation \(P \upsilon=R T\) is in error by less than \(5 \%\) when \(P / P_{\mathrm{c}}<0.05\) and \(T / T_{\mathrm{c}}>0.75\). To check this, compare these ratios for water, nitrogen, and helium at room conditions, say \(300 \mathrm{~K}\) and 1 bar. Which do you usually think of as gases at room conditions?


