8.2: Direction Liquefaction of Biomass
- Page ID
- 48593
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)8.2 Direction Liquefaction of Biomass
Credit: Dr. Caroline B. Clifford
There are differences for each of the thermal processes, as described in Lesson 5. Here we focus on direct liquefaction. Direct liquefaction (particularly hydrothermal processing) occurs in a non-oxidative atmosphere, where the biomass is fed into a unit as an aqueous slurry at lower temperatures, with bio-crude in the liquid form being the product. The primary focus of these particular processes is to produce a liquid product that is a hydrocarbon with an atomic H:C ratio of ~2, and a boiling range of 170-280 °C.
Many of the processes developed are based on coal-to-liquids processing. The main purposes in taking coal and biomass into a liquid is to produce liquids, to remove some of the less desirable components (i.e., sulfur, oxygen, nitrogen, minerals), and to make a higher energy density material that will flow.
One of the primary processes to convert coal into liquids directly is through a combination of thermal decomposition and hydrogenation under pressure. There are several single and two-stage processes that have been developed but have not been made commercial in the US. However, China opened a commercial direct liquefaction plant partially based on US designs in 2008. Figure 8.3a shows the general schematic of the plant as well as the products they make in Figure 8.3b. Design considerations include: 1) temperatures of ~400-450°C, 2) hydrogenation catalysts, 3) hydrocarbon solvents that are similar to fuels, 4) naturally occurring aromatics in coal, 5) sulfur, nitrogen, minerals that must be removed in refining of the liquid. Biomass can be processed in a similar manner, but biomass has significantly more oxygen and less aromatic compounds and decomposes differently than coal. Other processes have been developed for biomass, that appear to do a better job of processing cellulose. One process is hydrothermal processing in pressurized water using an acid catalyst such as LaCl3 at 250°C - we won't go into more detail here, but it is different from the direct liquefaction discussion in the next paragraph.
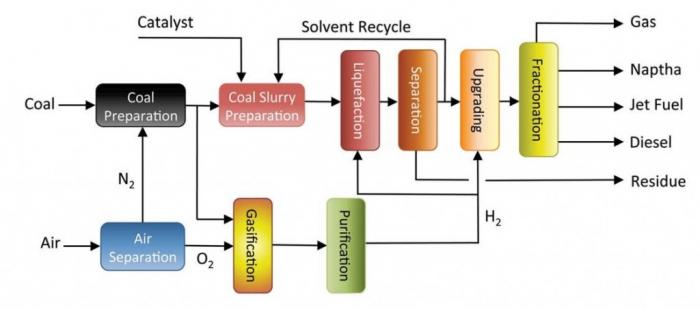
- Click Here for a text description of figure 8.3a
-
Schematic of Direct Coal Liquefaction.
Air enters the system and goes through air separation. This produces N2 and O2. N2 goes towards coal preparation and O2 going to gasification and purification.
The coal enters the system and goes to coal preparation. Follow preparation it can either go to gasification and then on to purification and liquefaction or upgrading. Or it can go into a coal slurry preparation process. This process requires a catalyst and a solvent. Following treatment, the slurry begins liquefaction, followed by separation. Following separation, the residue is removed, and the solvent is recycled. The rest of the product goes to upgrading where H2 is introduced. From upgrading the coal goes to fractionation which yields gas, naptha, jet fuel, and diesel.

Credit: Cornerstone
So, what are the differences with direct liquefaction of biomass? On the surface, it looks pretty much the same as the process for coal liquefaction. It is a thermochemical conversion process of organic material into liquid bio-crude and co-products. Depending on the process, it is usually conducted under moderate temperatures (300-400°C, lower than coal liquefaction) and pressures (10-20 MPa, similar or maybe a little higher with primarily hydrogen in coal to liquids) with added hydrogen or CO as a reducing agent. Unlike coal, the biomass is “wet”, or at least wetter than coal, and can be processed as an aqueous slurry. When processed as an aqueous slurry, the process is referred to in the literature as hydrothermal processing and can be subcritical to supercritical for water. Figure 8.4 shows the conditions for supercritical water; water behaves more like an acid/base system under these conditions. Thus, it can also be a catalyst. There is also a high solubility of organic material in water under these conditions. This mainly occurs along the liquid/vapor line. The basic reaction mechanisms can be described as:
- depolymerization of biomass;
- decomposition of biomass monomers by cleavage, dehydration, decarboxylation, and deamination;
- recombination of reactive fragments.
Different types of biomasses react differently depending on the biomass source. Carbohydrates, such as cellulose, hemicellulose, and starch can decompose in hydrothermal water. The typical product formed under these conditions is glucose, and glucose can then be fermented to make alcohol or further degrade in water to make glycolaldehyde, glyceraldehyde, and dihydroxyacetone. The products made depend on the conditions: at temperatures, ~180°C, products are sugar monomers, but at higher temperatures, 360-420°C, the aldehyde, and acetone compounds are formed.
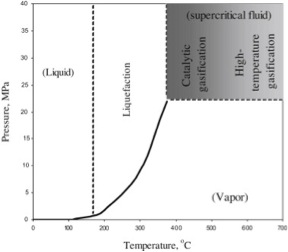
Credit: DOI: 10.1016/j.energy.2011.03.013
Lignin and fatty acids also decompose in hydrothermal water, but the products are very different because the substrate is different. For lignin, the products are similar to the building blocks for lignin, as shown in Figure 5.20a of Lesson 5 (p-coumaryl, coniferyl, and sinapyl alcohols), although the functional groups vary depending on the hydrothermal conditions. Bembenic and Clifford used hydrothermal water at 365°C and ~13 MPa to form methoxy phenols, using different gases to change the product slate (hydrogen, carbon monoxide, carbon dioxide, and nitrogen). For lipid or triglyceride (fats and oils) reaction in hydrothermal water at 330-340°C and 13.1 MPa, the main products are the free fatty acids (HC – COOH) and glycerol (C3H8O3). The free fatty acids can then be reacted to straight-chain hydrocarbons that can be used for diesel or jet fuel, although the temperature usually needs to be a little higher (400°C) for this to take place. Figure 8.5 shows the schematic of a hydrothermal water process to convert algae into liquid fuels, making use of heat from an integrated heat and power system. Flue gas from a power generation facility is used to grow algae. Algae is then harvested and concentrated in water. The algae are then reacted in a hydrothermal unit followed by catalytic hydrogenation to make the straight-chain hydrocarbon liquid fuels.
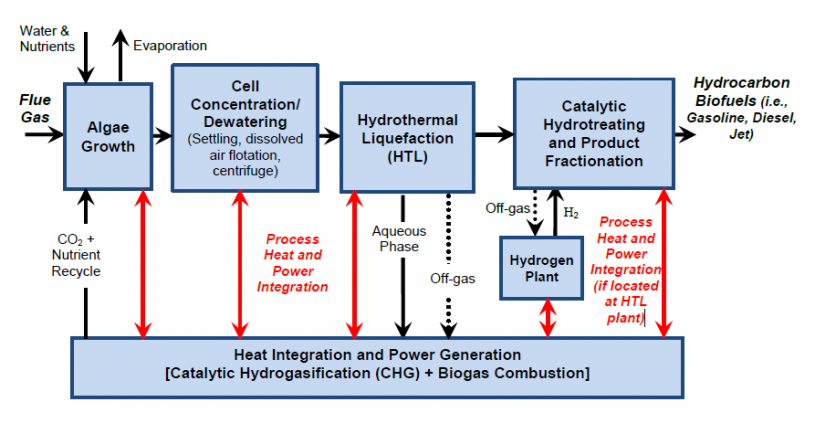
- Click Here for a text description of figure 8.5
-
Schematic for hydrothermal liquefaction of algae. First, the algae are grown, flue gas enters along with water, nutrients and recycled CO2. Evaporation occurs during this period. After growth, the next step is cell concentration/dewatering (settling, dissolved air flotation and centrifugation). Next is hydrothermal liquefaction (HTL) and the aqueous phase is removed. Finally, comes catalytic is hydrotreating and product fractionation. Here hydrogen gas from a hydrogen plant is added. This process yields hydrocarbon biofuels. In the diagram, all of these steps have double-headed arrows pointing to heat integration and power generation (catalytic hydrogasification (CHG) + Biogas Combustion).
Credit: USDE OSTI
Many types of catalysts can be used, although it depends on the process stage in which catalysts are used and what feed material is used. In hydrothermal processing, the more common catalysts used are acid and base catalysts. Particle size for biomass needs to be fine, with a size of < 0.5 mm. The introduction of the feed into the reactor is also challenging, as it is fed into a high-pressure reactor. Some advantages of using this process for biomass: 1) it is possible to process feeds with high water content, as much as 90%, 2) it is possible to process many different types of waste materials, including MSW, food processing waste, and animal manure, and 3) the process serves the dual roles of waste treatment and renewable energy production.
Process parameters include solids content, temperature, pressure, residence time, and use of catalysts. Often simultaneous reactions are taking place, which makes the overall understanding of the reactions complicated. The types of reactions taking place include solubilization, depolymerization, decarboxylation, hydrogenation, condensation, and hydrogenolysis.
For one particular process, hydrothermal liquefaction requires the use of catalysts. One typical catalyst used is sodium carbonate combined with water and CO to produce sodium formate:
Na2CO3 + H2O + CO → 2HCO2Na + CO2
This dehydrates the hydroxyl groups to carbonyl compounds, then reduces the carbonyl group to an alcohol:
HCO2Na + C6H10O5 → C6H10H4 + NaHCO3
H2 + C6H10O5 → C6H10H4 + H2O
The formate and hydrogen can be regenerated and recycled. Other catalysts used that behave in a similar manner include K2CO3, KOH, NaOH, and other bases. For simultaneous decomposition and hydrogenation, nickel (Ni) catalysts are used.
Similar to pyrolysis, the major product of this process is a liquid biocrude, which is a viscous dark tar or asphalt material. Up to 70% of the carbon is converted into biocrude; lighter products are obtained when different catalysts are used. Co-products include gases (CO2, CH4, and light hydrocarbons) as well as water-soluble materials. The liquid biofuel has a similar carbon to hydrogen ratio as in the original feedstock and is a complex mixture of aromatics, aromatic oligomers, and other hydrocarbons. In this process, the oxygen is reduced and is 10-20% less than typical pyrolysis oils, with a heating value higher than pyrolysis oils, 35-40 MJ/kg on a dry basis. However, the USDA has developed a pyrolysis process using recycled gases that produces a fairly light hydrocarbon with very little oxygen content. (Mullens et al.) I will discuss this more in the next section. Table 8.2 shows a comparison of biocrudes from various processes and feed materials. The quality of the biocrude shown from hydrothermal processing is for a heavy biocrude. Other processes will make a lighter material, but also produce more co-products that must be utilized as well.
Table 8.2: Comparison of biocrude from hydrothermal processing, bio-oil from fast pyrolysis, and heavy petroleum fuel.
| Characteristic | Hydrothermal Bio-oil | Fast pyrolysis Bio-oil | Heavy Petroleum Fuel | USDA Oil Oak | ||
|---|---|---|---|---|---|---|
| Water Content, wt% | 3-5 | 15-25 | 0.1 | 4.8 | ||
| Insoluble solids, % | 1 | 0.5-0.8 | 0.01 | n/a | ||
| HHV, MJ/kg | 30 | 17 | 40 | 34.0 | ||
| Density, g/ml | 1.10 | 1.23 | 0.94 | n/a | ||
| Viscosity, cp | 3,000-17,000 | 10-150 | 180 | n/a | ||
| Wet | Dry | Wet | Dry | |||
| Carbon, % | 73.0 | 77.0 | 39.5 | 55.8 | 85.2 | 80.2 |
| Hydrogen, % | 8.0 | 7.8 | 7.5 | 6.1 | 11.1 | 5.9 |
| Oxygen, % | 16.0 | 13.0 | 52.6 | 37.9 | 1.0 | 11.8 |
| Nitrogen, % | <0.1 | <0.1 | <0.1 | <0.1 | 0.3 | 2.1 |
| Sulfur, % | <0.05 | <0.5 | <0.05 | <0.5 | 2.3 | n/a |
| Ash, % | 0.3-0.5 | 0.3-0.5 | 0.3-0.5 | 0.2-0.3 | <0.1 | n/a |


