14.12: Thermodynamics (Statistical Physics)
- Page ID
- 45592
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Thermodynamic Definitions
The previous section dealt with energy in the mechanical world, but not heat energy (or electric). Thermodynamics is the disciplines that deals with heat and its relation to other forms of energy. It is part of the overall discipline of statistical mechanics. To start off with this brief introduction to thermodynamics we will define some essential terms.
| Property | In a system this is a quantity (measurable) that is to be described (like pressure or volume) |
| State | Properties at a given instance |
| Process | This is the path of successive states through the application of energy (heat, mechanical, etc.) |
| Thermodynamic Equilibrium | When the properties of a system have ceased to change AND have no tendency to change |
| Metastable | When the properties of the system are seemingly stable but have a tendency to change |
iso's
|
When a property is constant You will normally see this terms used when discussing a P-V diagram or one of the other related diagrams |
| Adiabatic | Heat does not enter or leave a system |
| Phase | When the properties of a system are uniform |
| Phase transition |
When a system goes from one uniform property system to another This is associated with a change in the degrees of freedom |
| Entropy | A measure of the disorder in a system |
| Enthalpy | A measure of the heat of a system |
| Free energy | Internal energy that is usable for work (hence it is "free" to use) |
| Helmholtz free energy |
Entropy-based free energy which assumes constant temperature and volume Normally favored by Physicists |
| Gibbs free energy |
Enthalpy-based free energy which assumes a constant pressure Normally favored by Chemists |
| Partition function |
A function that describes statistically a thermodynamic equilibrium system in terms of thermodynamic properties Statistical mechanics |
Statistical Mechanics
In physics thermodynamics is derived from the overall subject of statistical mechanics. Statistical mechanics uses statistical methods to describe systems of microscopic elements in terms of macro concepts like temperature and pressure. Temperature and pressure do not exist as far as single particles is concerned because they are statistical concepts. So in physics statistical is used to describe these concepts. For thermodynamics the most important concept is the partition function. The partition function is dimensionless entity that is express as
\[Z \equiv \sum_r {e^{-\beta E_r}}\]
where \(E_r\) is the total energy in each state r and \(\beta\) is the thermodynamic beta defined as \(\frac{1}{kT}\) where k is the Boltzman's constant. The thermodynamic beta represents coldness. A little bit of heat added will increase entropy with is expressed by the "less" coldness of the thermodynamic beta. Thus the thermodynamic beta is directly related to entropy.
Most of the properties of thermodynamics can be derived from the partition function. Statistical mechanics is a graduated level physics course and as such we will not go any further on this subject, but instead move on to more concrete thermodynamics.
Entropy
Entropy is a measure of thermal energy not convertible into work. This is better understood as the disorder of a system as work is derivable from ordered state motion. The more disorder the higher the entropy and the less work available from thermal sources. We can express entropy as
\[S = k \ln \Omega\]
where \(\Omega\) is the number of states in a system (for an ideal gas you would have \(\Omega \propto V^N E^{3N/2}\) were V is volume, E is energy, and N is the number of molecules).
Laws of Thermodynamics
| 0th Law | If TA = TB and TB = TC then TA = TC |
| 1st Law | \(Q - W = \Delta U\) where Q is thermal energy (heat), W is work, and U is internal energy |
| 2nd Law | Stotal cannot decrease (note however local entropy can decrease...) |
| 3rd Law | As \(T \rightarrow 0 K\) then \(S \rightarrow 0\) \(\Rightarrow\) \(\therefore\) can't reach absolute zero |
Equations of some thermodynamic properties
There are many properties in thermodynamics but there are four properties that are generally very familiar: Volume, Pressure, Temperature, and mass density. Volume is mostly obvious from mathematics class. Pressure is \(P= \frac{\vec{F_n}}{A}\) where \(F_n\) is the normal force on a surface and A is the cross-sectional area. However this is not in terms of a thermodynamic system because the force and area are a macro concept, not a micro concept, so we it might be better to define pressure as \(P = - \left( \frac{\partial U}{\partial V} \right)_{S,N}\) which states that pressure is the change of internal energy with respect to changing volume with entropy being held constant (and implicitly with the number of states not changing). Mass density is \(\rho = \frac{m}{V}\) where m is mass and V is volume. Temperature1 like pressure need to be expressed with a differential equation because of it is a micro concepts (atoms, molecules, particles). \(T = \left( \frac{\partial U}{\partial S} \right)_{V,N}\) which states that temperature is the change of internal energy with respect to changing entropy with volume being held constant (and implicitly with the number of states not changing).
Ideal Gas Law (and less ideal gas law)
The ideal gas law is an equation that relates pressure, volume, and temperature with an assumption that the system is ideal (no interaction between the molecular particles). It is a fairly good approximate in a lot of situations. The equation is
\[PV = nRT\]
where P is pressure, V is volume, n is the number of moles, R is the universal gas constant, and T is temperature. This law is an accumulation of different laws like Boyle's Law (Mariotte's Law) and Charles's Law.
For a sort of non-ideal gas law we will start with the ideal gas law and modify it. The ideal gas law equation can also be expressed using a specific gas constant, \(\overline{R}\) using mass (m).
\[PV = m \overline{R} T\]
If we use specific volume we can rewrite this equation as
\[P \nu = \overline{R} T\]
It is this equation where we can add a constant to that will allow
\[P \nu = Z \overline{R} T\]
where Z is a compressibility factor. For Z = 1 we have the ideal equation again and for \(Z \neq 1\) we have an expression for a non-ideal situation. There is more to this story but in this brief introduction we will move on to the next subject which is a breakdown of the first law starting with work and its many forms.
The First Law
Work equations
In order to get a better understanding of work we list some differential work expressions with their "origin." This is not a complete list or any sort of extensive proof of concept. This is just for you all to get a handle on the idea of work after which we will discuss heat (flow) and then internal energy.
| Differential work equation | Details | Type of work |
|---|---|---|
| dW = F ds | F is force, s is distance | General work (due to force) - kinetic energy |
| dW = P dV | P is pressure, V is volume | Expansion work |
| dW = mg dh | m is mass, g is gravity, h is height | Gravitational work - potential energy |
| dW = V dq | V is voltage, q is charge | Electrical work |
| dW = \(\mu\) dn | \(\mu\) is chemical potential, n is number of moles | Chemical work |
| dW = \(\gamma\) dA | \(\gamma\) is surface tension, A is surface area | "Surface tension" work |
| dW = k dx | k is spring constant, x is "stretching" of spring (or springy thing) | Spring work (or stretching work) |
Heat (flow)
Generally when we talk about heat we start with the heat equation which is a parabolic partial differential equation which is rather complex at this stage of your academic career. To discuss heat we will instead go to the historical and still valid ideas of Newton and Fourier.2
So in the following table we present the equations on heat flow.
| Law | Uses | Equation |
|---|---|---|
| Newton's Law | Convection (fluid) | \(\dot{Q} = hA\Delta T = \frac{A \Delta T}{r}\) |
| Fourier's Law (1D) | Conduction (molecules/materials) | \(\dot{Q} = \frac{kA\Delta T}{L} = \frac{A\Delta T}{r}\) |
| Fourier's Law (3D) | Conduction/heat flux | \(\vec{\phi_q} = -k \nabla T\) |
| Stefan-Boltzmann Law | Radiation | \(\dot{Q} = \sigma \epsilon A (T_1^4 - T_2^4)F_{1-2}\) |
where h is heat transfer coefficient, A is surface area where heat transfer is going through, r is resistivity factor, k is thermal conductivity, \(\vec{\phi_q}\) is heat flux which using the heat equation can be reformulated in to heat flow which is done for one dimension above, \(\sigma\) is Stefan-Boltzmann3 constant \(\sigma=5.670374 \times 10^{-8} W \cdot m^{-2} \cdot K^{-4}\), \(\epsilon\) is emissivity (one for a black body), and \(F_{1-2}\) is a shape factor.
Integrating the heat flow4 over time will give the heat energy.
Internal Energy
Internal energy, U, is the energy that is transferred either through heat or mass internally in a system. It is not measurable except through other measurable energies. Internal energy is dependent on entropy, S, volume, V, and the particles. Internal energy does not depend on the kinetic and potential of the system as a whole, but you could view it as the kinetic and potential energy of the microscopic particles. That viewpoint will not work in the relativistic point of view however.
Carnot Cycle (Ideal Engine)
To complete this introduction to thermodynamics5 we will briefly go over the Carnot cycle which describes the heat-work relationship particularly in engines or cryocoolers (reverse Carnot cycle). The Carnot cycles is the theoretical ideal thermal-work cycle (engine). Carnot cycle cannot be achieved but is the goal that all other cycles seek (Stirling, Otto, etc.).
Carnot cycles are normally described on a graph two different ways: Pressure versus Volume and Temperature versus Entropy.
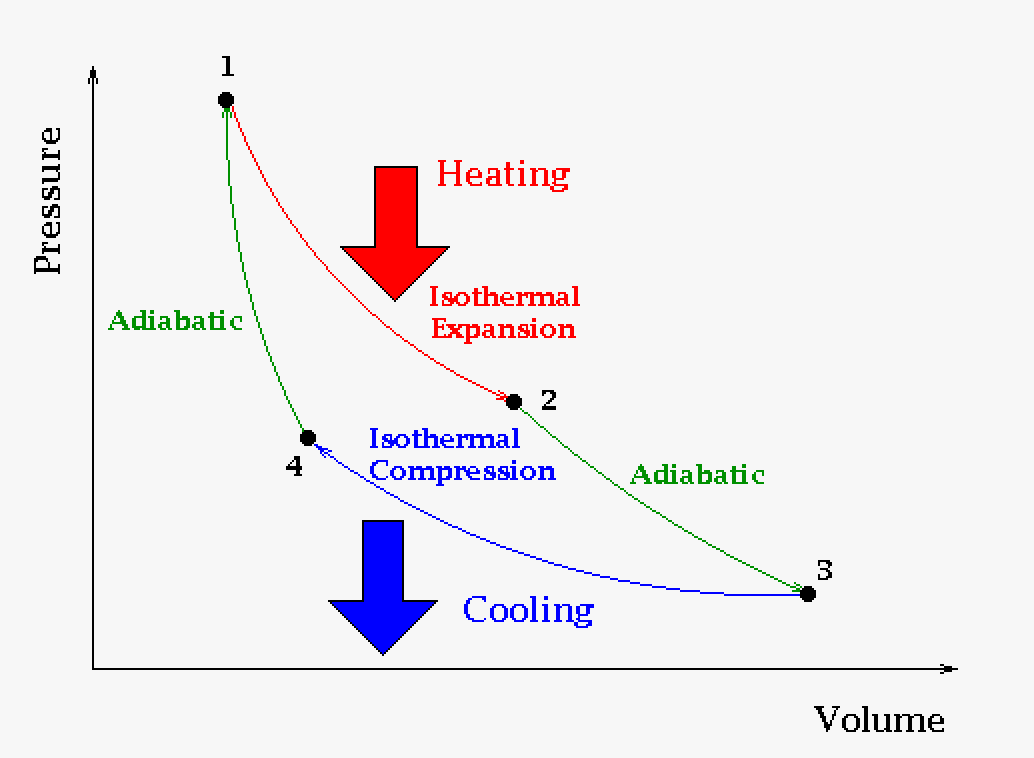 |
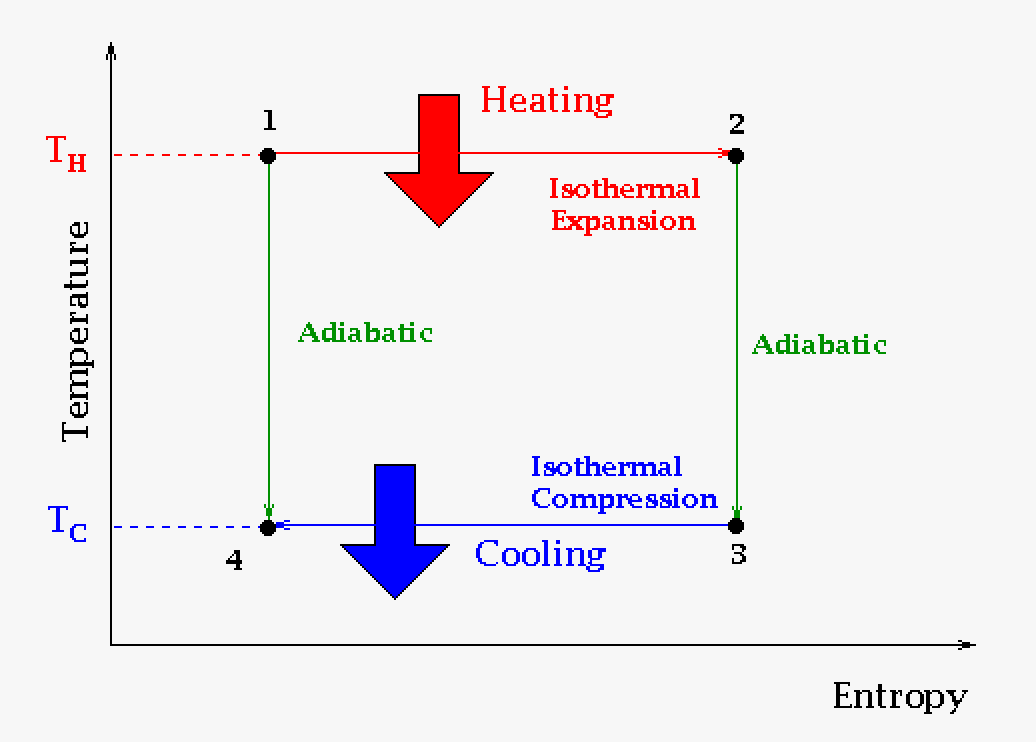 |
|
The pressure-volume description of the engine cycle is used most commonly. The above graph is of the Carnot cycle. When we apply a heat source at 1 to a system that is free to expand we go through an isothermal expansion as it absorbs heat and the volume increase as a result (pressure decreases). The heat source is removed at 2 and the system goes through an adiabatic expansion. Next we apply a cold source at 3 to the system and it goes through a isothermal compression and the volume decreases as a result. The cold source is removed at 4 and the system goes through an adiabatic compression. The work done is the area in the enclosed cycle. |
The temperature-entropy description of the engine cycle is used less commonly. The main reason to use this is to see the change in entropy. Note that the ideal process (the Carnot cycle) does not change the entropy (universal). Basically the "entropy source" is equal to the "entropy sink." Real world cycles like the Otto or Stirling cycle will not see this behavior (though they will get as close as they can). Basically the amount of useable heat to produce work will all go towards producing work in the Carnot cycle. |
Real world cycles will modify the Carnot cycle such as putting heat and work into the system (Otto cycle) to get more work out. Some cycles are isochoric so that clearly also will modify the Carnot cycle.
Reverse Carnot Cycle (Cryocoolers)
If we reverse the Carnot cycle we have a description of how refrigerators or cryocoolers work. The Stirling cycle is one of the most common cycles reversed to produce the Stirling cryocooler (as you might have noted when doing your laboratory design project the Stirling cyrocooler was efficient, but produced a lot of vibration).
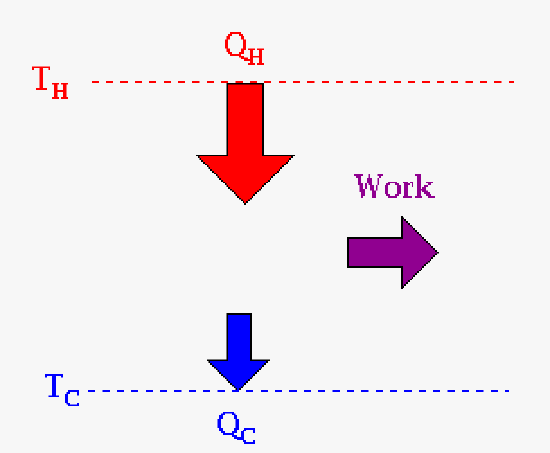 |
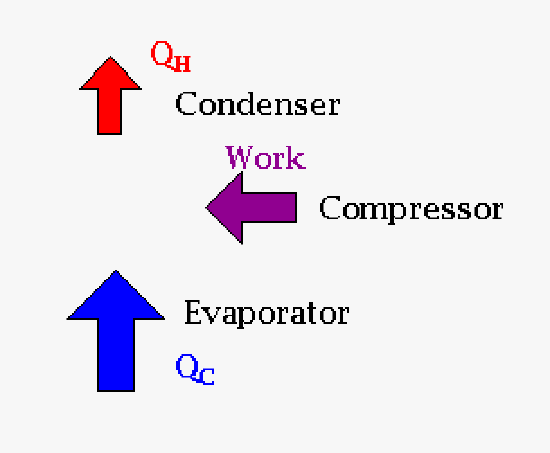 |
| The relationship between heat and work for an engine (Carnot cycle). The engineering result that a customer would want is work. | The relationship between heat and work for a cryocooler. Here work is done to produce a cold surface that you might place your detector on to cool it. Why? If you remember from the laboratory design it has something to do with noise on your detector. |
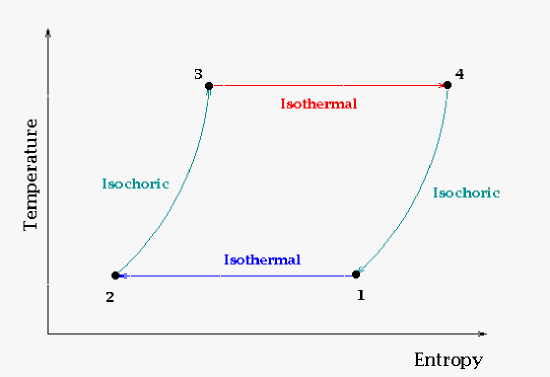 |
| Stirling cycle as presented using the temperature-entropy graph. Here you will note the volume is constant. |
Going back to a previous section, the laboratory design, you should have investigated the idea of a cryocooler (and compared it to a cryostat (cryo-Dewar) and TECs). Here is a simple outline of cryocoolers with a description of a real world issue that is not discussed in thermodynamics: Vibration.
- Stirling Engine (Stirling Cryocooler)
- Repeated cooling and heating of a sealed amount of working gas
- Air, hydrogen, helium
- Follows standard gas law behavior PV=nRT
- Essentially same theoretical efficiency as Carnot engine
- Stirling engines can be reversed to make a Stirling cryocooler
- High vibration
- Repeated cooling and heating of a sealed amount of working gas
- Gifford-McMahon Cryocoolers
- Version of a Stirling cryocooler
- Inexpensive
- High vibration
- Vuilleumier Cryocoolers
- Version of a Stirling cryocooler
- Mechanically linked two displacers (different from Stirling)
- The displacer is a special "piston" in certain Stirling-type cryocoolers whose purpose is to move the working gas back and forth between the hot and cold exchangers.
- The displacer is not the main piston(s) that is normally attached to a flywheel.
- The classic Stirling cryocooler does not have a displacer.
- Low noise
- Reliable (as are all Stirling-type cryocoolers)
- Joule-Thomson Cryocoolers
- Throttling process
- Moisture problem in the throttling process is a known problem. Mini-JT cryocoolers might suffer from this problem worse.
- Low vibration
- Low efficiency
- Usually combined with other cryocooler technology
- Can be operated without other cryocoolers, but need special compressor
- Sorption cryocooler
- Type of Joule-Thomson cryocooler
- Achieves the thermodynamics through a chemical means (adsorption).
- Low efficiency
- Very low vibration
- Brayton Engine (Brayton Cryocooler)
- Turbine-based compressor-type engines
- Can be reversed to produce a Brayton cryocooler
- Highly efficient at low temperatures
- HST's NICMOS uses a turbo-Brayton cryocooler uses balanced turbines to achieve vibration-free performance
- Flourescent cryocooler
- Solid state cryocooler
- No moving parts as in Stirling-type cryocoolers
- Low mass
- New technology still being developed but could be promising.
- Pulse-Tube Cyrocoolers
- Using a compressor on a regenerator connected to a sealed tube produces a cold end and a warm end
- Efficiency can be increased by changing manipulating the mass oscillation (see Orifice Pulse Tube handout)
- Benefits
- No cold moving parts as in Stirling-type cryocoolers (which will have vibrational benefits, of course)
- Low vibration (mostly caused by the compressor which can be isolated)
- High efficiency
- Inexpensive
- Drawbacks
- Compressor, usually very big and noisy
- Cooling of compressor either by water or air
- Large for space applications, but versions for space have been achieved
- Miniature pulse-tubes might suffer from boundary condition effects with smaller and smaller tubes. This will decrease their efficiency.
Liquid cryostat ("Dewars")
Cryocoolers (non-liquid cryostats) are to be compared to liquid cryostats (in the sense of "scientific instrument Dewars") and Thermoelectric Coolers (TECs). Cryocoolers main drawback is vibration where as Dewars and TECs have essentially zero vibration though TECs may have a thermal footprint as any electronic device might have especially in the scientific arena.
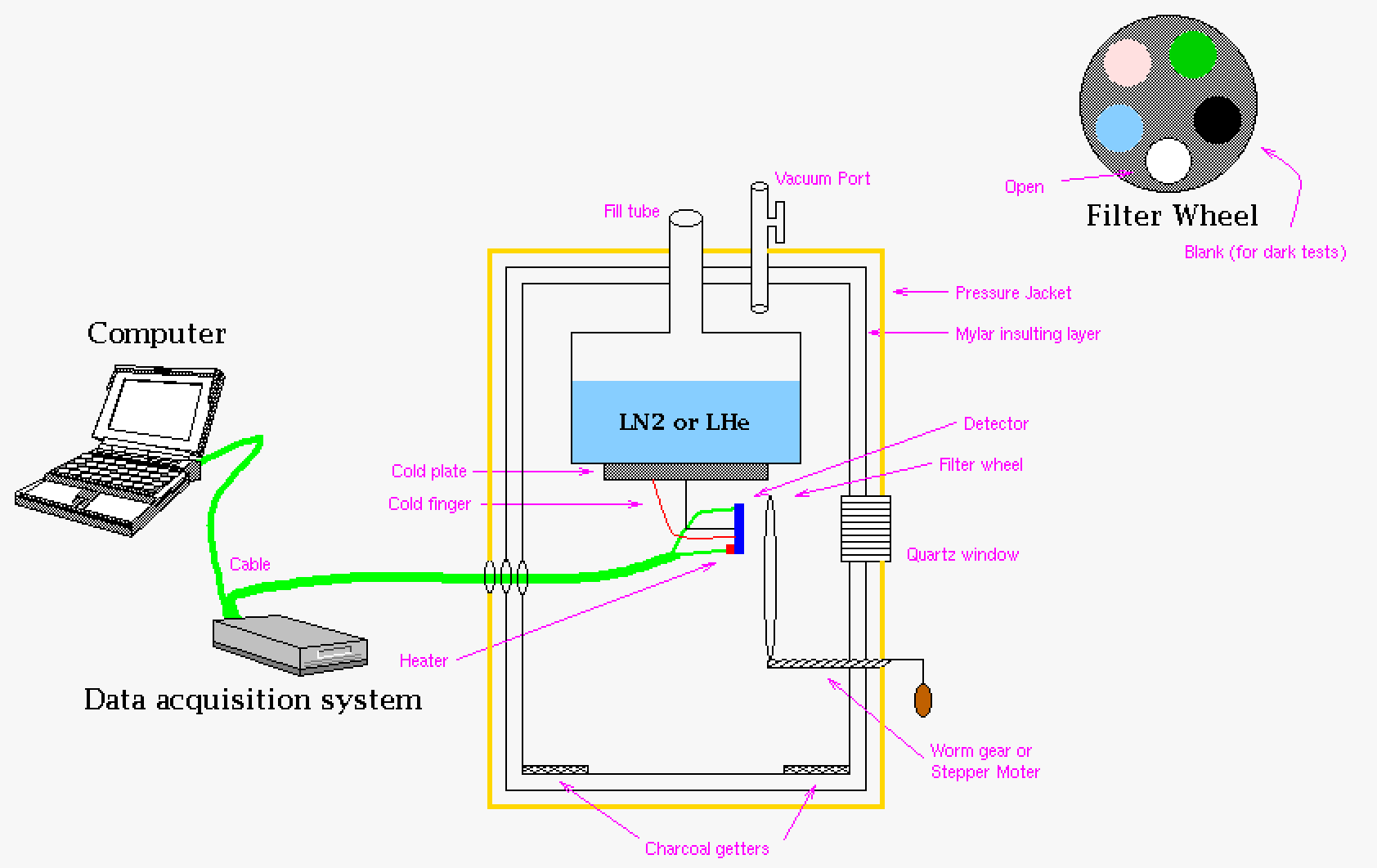 |
| A cartoon of a Dewar (liquid cryostat) for the propose of cooling detectors. Pictured here are the items usually associated with running a detector such as a data acquisition system and computer. In the Dewar is the detector and a filter wheel that is moved by a worm gear (see gears in Solid Mechanics section or just do a quick search on the web). Because the detector is cooled there are getters to take up any residual moisture in the vacuum chamber. This image is exaggerated in order to label items within a Dewar system (note there are many many variations on this idea). |
Using the Dewar figure above we could look at the various heat flow laws in relationship to preventing heat flow into the cooled chamber where the detector is mounted and tested in the system. This is instructive cyrogenic systems.
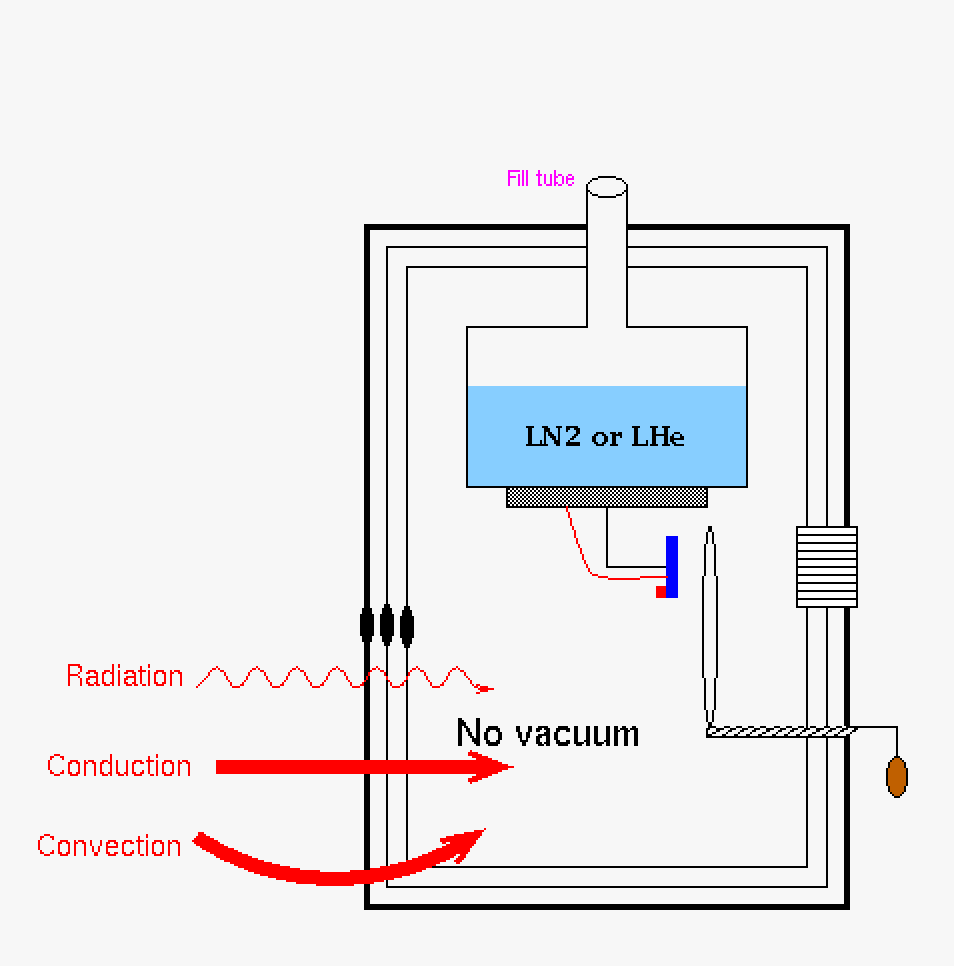 |
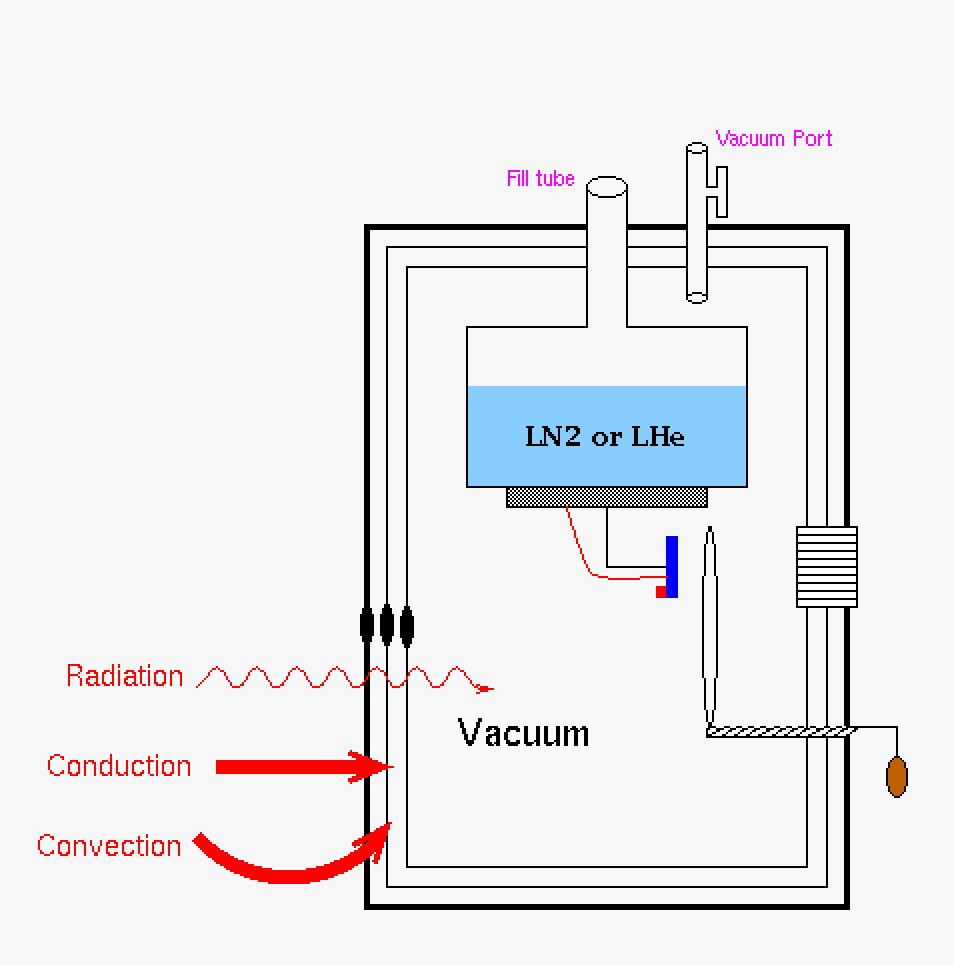 |
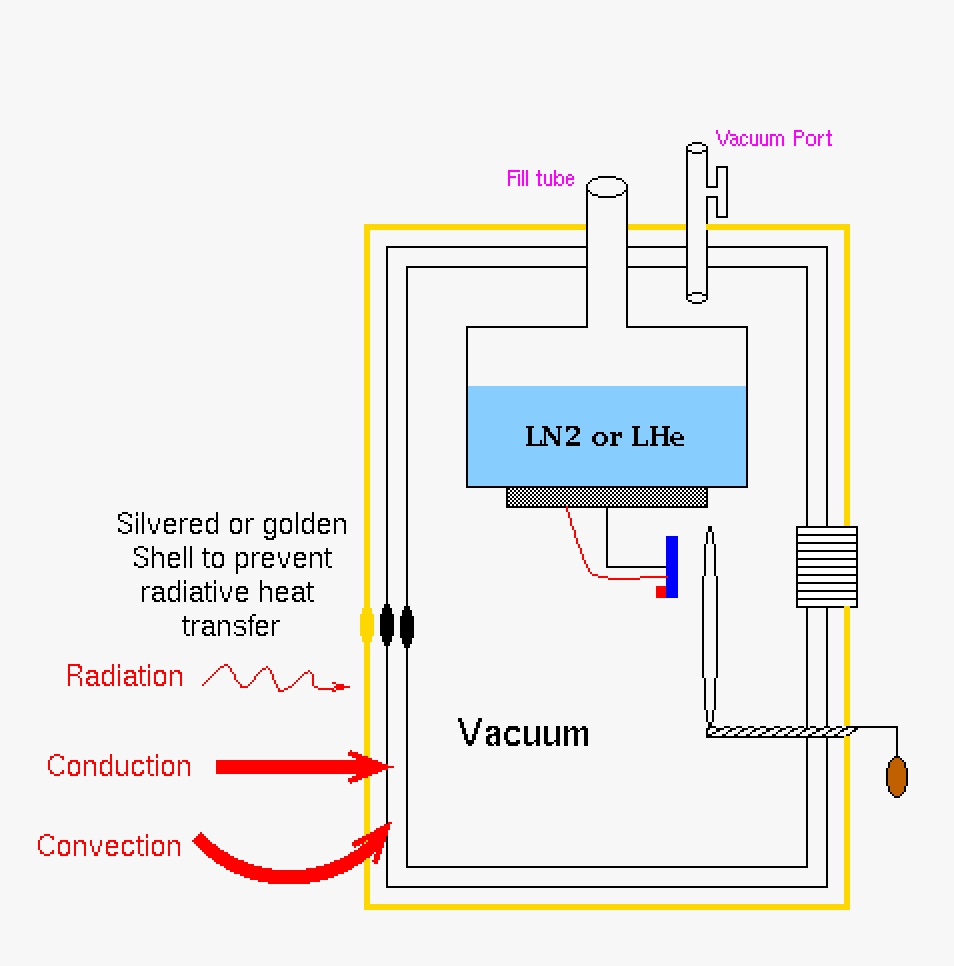 |
| This is our Dewar with no vacuum and a non-silvered (or similar cladding) jacket. All mechanisms of heat transfer will cause the system to heat up. This is not a good design. | Pumping down the Dewar through the vacuum port allows for a vacuum chamber where the detector is mounted in the system. Conduction and convection heat flow is stymied, however radiation heat transfer is still getting into the system. | Putting a silvered (or similar cladding) jacket on the Dewar will prevent radiation heat transfer. This is the best configuration for a Dewar. |
That completes this brief journey into thermodynamics6 (and applications) for this course. You will learn more about thermodynamics and the thermodynamic cycle in physics and possibly a thermodynamics course (should take if you are interested, of course).
1A more intuitive way but not reliable way of looking at temperature is by using kinetic theory to used average molecular kinetic energy and the ideal gas law to derive a so-called kinetic temperature. While more intuitive it is not accurate in a number of situations.
2Joseph Fourier, the mathematician behind the Fourier series and Fourier transform, also was a physicist who worked on a theory of heat flow that was based of Newton's Law of Cooling. From these formulas comes a the more complex heat equation which we will not cover here. A side note, Fourier is the first to figure out the greenhouse effect.
3If on a test you were given this constant but not the equation you could use the units of this constant to help you remember that radiative heat flow should have T4 rather then T in the equation. Hint, hint.
4We use heat flow here because heat transfer is implied in heat, Q, itself. \(\dot{Q}\), which is the time derivative of heat, is really then the flow of heat transfer of time. However engineers call this quantity heat transfer which is like saying heat transfer transfer not unlike saying ATM machine or SAT test. We believe heat flow is more expressive.
5The ideas in this section of apply heating and cooling for the purpose of producing engines is where the term thermodynamics came from. This process is clearly dynamic and is the counter to the debate that thermodynamics is actually thermostatics.
6It should be noted that while in modern times thermodynamics is derived from statistical mechanics, historically thermodynamics was developed first. Statistical mechanics was developed to give thermodynamics a theoretical underpinning.

